| UTHEALTH HOME ABOUT SBMI A-Z WEBMAIL INSIDE THE UNIVERSITY |

|
|||||||
|
Kinase Fusion Gene:AKT2_AKT2 |
Kinase Fusion Protein Summary |
 Kinase Fusion gene summary Kinase Fusion gene summary |
| Kinase Fusion partner gene information | Kinase Fusion gene name: AKT2_AKT2 | KinaseFusionDB ID: KFG269 | FusionGDB2.0 ID: KFG269 | Hgene | Tgene | Gene symbol | AKT2 | AKT2 | Gene ID | 208 | 208 | |
| Gene name | AKT serine/threonine kinase 2 | AKT serine/threonine kinase 2 | ||||||||||
| Synonyms | HIHGHH|PKBB|PKBBETA|PRKBB|RAC-BETA | HIHGHH|PKBB|PKBBETA|PRKBB|RAC-BETA | ||||||||||
| Cytomap | 19q13.2 | 19q13.2 | ||||||||||
| Type of gene | protein-coding | protein-coding | ||||||||||
| Description | RAC-beta serine/threonine-protein kinasePKB betaRAC-PK-betamurine thymoma viral (v-akt) homolog-2protein kinase Akt-2protein kinase B betaputative v-akt murine thymoma viral oncoprotein 2rac protein kinase betav-akt murine thymoma viral oncogene h | RAC-beta serine/threonine-protein kinasePKB betaRAC-PK-betamurine thymoma viral (v-akt) homolog-2protein kinase Akt-2protein kinase B betaputative v-akt murine thymoma viral oncoprotein 2rac protein kinase betav-akt murine thymoma viral oncogene h | ||||||||||
| Modification date | 20240413 | 20240413 | ||||||||||
| UniProtAcc | P31751 | P31751 | ||||||||||
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000311278, ENST00000392038, ENST00000424901, ENST00000579047, ENST00000581582, | ENST00000392038, ENST00000579047, ENST00000311278, ENST00000424901, ENST00000581582, | |||||||||
| Context (manual curation of fusion genes in KinaseFusionDB) | PubMed: AKT2 [Title/Abstract] AND AKT2 [Title/Abstract] AND fusion [Title/Abstract] | |||||||||||
| Most frequent breakpoint (based on all fusion genes of FusionGDB 2.0) | AKT2(40740983)-AKT2(40791210), # samples:1 | |||||||||||
 Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez |
| Partner | Gene | GO ID | GO term | PubMed ID |
| Hgene | AKT2 | GO:0030335 | positive regulation of cell migration | 25428377 |
| Tgene | AKT2 | GO:0030335 | positive regulation of cell migration | 25428377 |
 Kinase Fusion gene breakpoints across AKT2 (5'-gene) Kinase Fusion gene breakpoints across AKT2 (5'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 Kinase Fusion gene breakpoints across AKT2 (3'-gene) Kinase Fusion gene breakpoints across AKT2 (3'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
Top |
Kinase Fusion Gene Sample Information |
 Kinase Fusion gene information. Kinase Fusion gene information. |
 Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)* All genome coordinats were lifted-over on hg19. * Click on the break point to see the gene structure around the break point region using the UCSC Genome Browser. |
| Source | Sample | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp |
| ChiTaRS5.0 | BF739868 | AKT2 | chr19 | 40740983 | AKT2 | chr19 | 40791210 |
Top |
Kinase Fusion ORF Analysis |
 Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. |
| Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | Seq length (transcript) | Seq length (amino acids) |
Top |
Kinase Fusion Amino Acid Sequences |
 For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. |
| >Henst_Tenst_Hgene_Hchr_Hbp_Tgene_Tchr_Tbp_length(fusion AA)_AAseq |
Multiple Sequence Alignment of All Fusion Protein Isoforms |
Top |
Kinase Fusion Protein Functional Features |
 Four levels of functional features of fusion genes Four levels of functional features of fusion genesGo to FGviewer search page for the most frequent breakpoint (https://ccsmweb.uth.edu/FGviewer/:40740983/:40791210) - FGviewer provides the online visualization of the retention search of the protein functional features across DNA, RNA, protein, and pathological levels. - How to search 1. Put your fusion gene symbol. 2. Press the tab key until there will be shown the breakpoint information filled. 4. Go down and press 'Search' tab twice. 4. Go down to have the hyperlink of the search result. 5. Click the hyperlink. 6. See the FGviewer result for your fusion gene. |
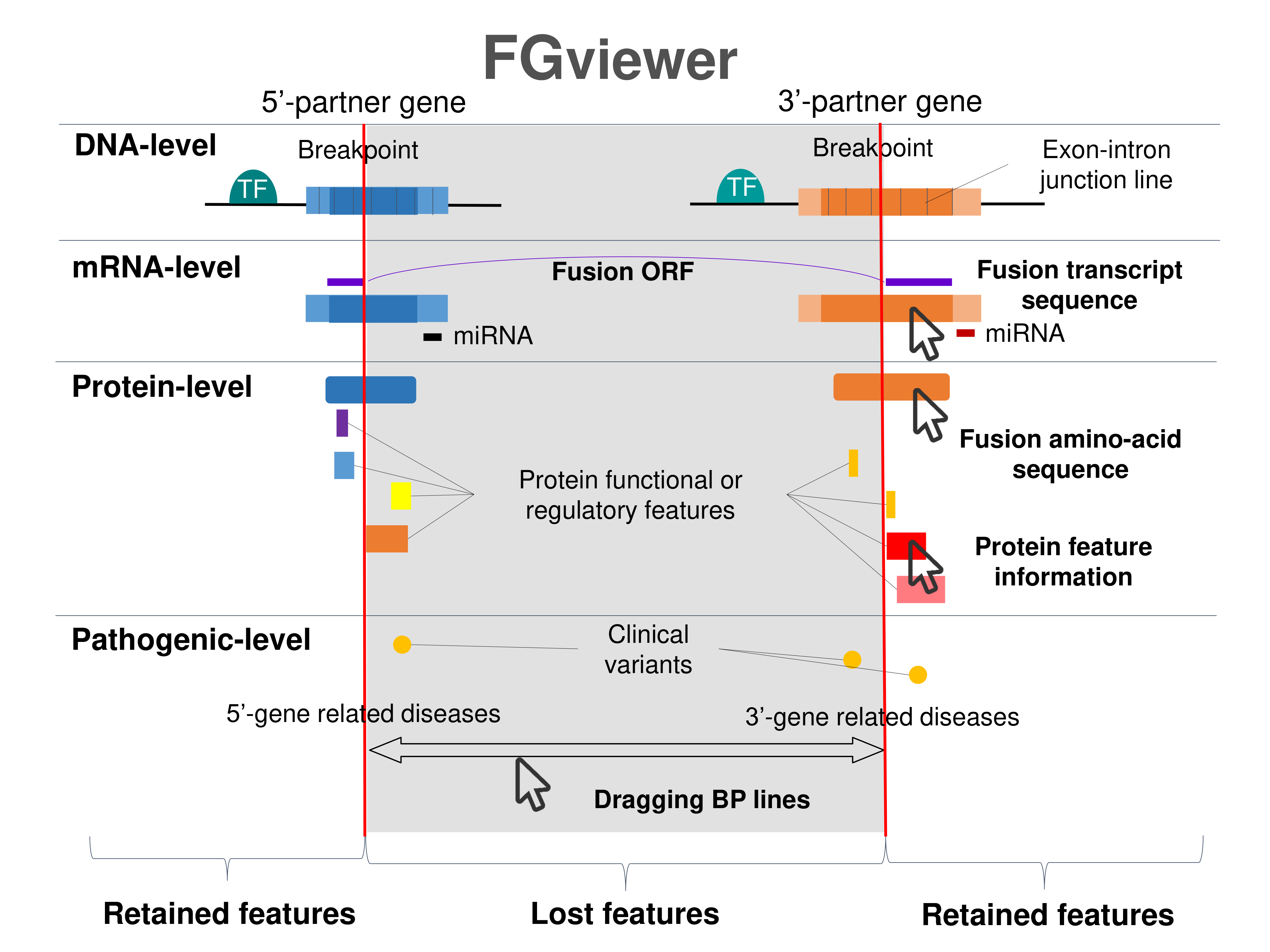 |
 Main function of each fusion partner protein. (from UniProt) Main function of each fusion partner protein. (from UniProt) |
| Hgene | Tgene |
| AKT2 | AKT2 |
| FUNCTION: AKT2 is one of 3 closely related serine/threonine-protein kinases (AKT1, AKT2 and AKT3) called the AKT kinase, and which regulate many processes including metabolism, proliferation, cell survival, growth and angiogenesis. This is mediated through serine and/or threonine phosphorylation of a range of downstream substrates. Over 100 substrate candidates have been reported so far, but for most of them, no isoform specificity has been reported. AKT is responsible of the regulation of glucose uptake by mediating insulin-induced translocation of the SLC2A4/GLUT4 glucose transporter to the cell surface. Phosphorylation of PTPN1 at 'Ser-50' negatively modulates its phosphatase activity preventing dephosphorylation of the insulin receptor and the attenuation of insulin signaling. Phosphorylation of TBC1D4 triggers the binding of this effector to inhibitory 14-3-3 proteins, which is required for insulin-stimulated glucose transport. AKT regulates also the storage of glucose in the form of glycogen by phosphorylating GSK3A at 'Ser-21' and GSK3B at 'Ser-9', resulting in inhibition of its kinase activity. Phosphorylation of GSK3 isoforms by AKT is also thought to be one mechanism by which cell proliferation is driven. AKT regulates also cell survival via the phosphorylation of MAP3K5 (apoptosis signal-related kinase). Phosphorylation of 'Ser-83' decreases MAP3K5 kinase activity stimulated by oxidative stress and thereby prevents apoptosis. AKT mediates insulin-stimulated protein synthesis by phosphorylating TSC2 at 'Ser-939' and 'Thr-1462', thereby activating mTORC1 signaling and leading to both phosphorylation of 4E-BP1 and in activation of RPS6KB1. AKT is involved in the phosphorylation of members of the FOXO factors (Forkhead family of transcription factors), leading to binding of 14-3-3 proteins and cytoplasmic localization. In particular, FOXO1 is phosphorylated at 'Thr-24', 'Ser-256' and 'Ser-319'. FOXO3 and FOXO4 are phosphorylated on equivalent sites. AKT has an important role in the regulation of NF-kappa-B-dependent gene transcription and positively regulates the activity of CREB1 (cyclic AMP (cAMP)-response element binding protein). The phosphorylation of CREB1 induces the binding of accessory proteins that are necessary for the transcription of pro-survival genes such as BCL2 and MCL1. AKT phosphorylates 'Ser-454' on ATP citrate lyase (ACLY), thereby potentially regulating ACLY activity and fatty acid synthesis. Activates the 3B isoform of cyclic nucleotide phosphodiesterase (PDE3B) via phosphorylation of 'Ser-273', resulting in reduced cyclic AMP levels and inhibition of lipolysis. Phosphorylates PIKFYVE on 'Ser-318', which results in increased PI(3)P-5 activity. The Rho GTPase-activating protein DLC1 is another substrate and its phosphorylation is implicated in the regulation cell proliferation and cell growth. AKT plays a role as key modulator of the AKT-mTOR signaling pathway controlling the tempo of the process of newborn neurons integration during adult neurogenesis, including correct neuron positioning, dendritic development and synapse formation. Signals downstream of phosphatidylinositol 3-kinase (PI(3)K) to mediate the effects of various growth factors such as platelet-derived growth factor (PDGF), epidermal growth factor (EGF), insulin and insulin-like growth factor I (IGF-I). AKT mediates the antiapoptotic effects of IGF-I. Essential for the SPATA13-mediated regulation of cell migration and adhesion assembly and disassembly. May be involved in the regulation of the placental development. Involved in the inhibition of ciliogenesis associated with RAB8-dependent cilia growth (PubMed:31204173). {ECO:0000269|PubMed:31204173}.; FUNCTION: One of the few specific substrates of AKT2 identified recently is PITX2. Phosphorylation of PITX2 impairs its association with the CCND1 mRNA-stabilizing complex thus shortening the half-life of CCND1. AKT2 seems also to be the principal isoform responsible of the regulation of glucose uptake. Phosphorylates C2CD5 on 'Ser-197' during insulin-stimulated adipocytes. AKT2 is also specifically involved in skeletal muscle differentiation, one of its substrates in this process being ANKRD2. Down-regulation by RNA interference reduces the expression of the phosphorylated form of BAD, resulting in the induction of caspase-dependent apoptosis. Phosphorylates CLK2 on 'Thr-343'. | FUNCTION: AKT2 is one of 3 closely related serine/threonine-protein kinases (AKT1, AKT2 and AKT3) called the AKT kinase, and which regulate many processes including metabolism, proliferation, cell survival, growth and angiogenesis. This is mediated through serine and/or threonine phosphorylation of a range of downstream substrates. Over 100 substrate candidates have been reported so far, but for most of them, no isoform specificity has been reported. AKT is responsible of the regulation of glucose uptake by mediating insulin-induced translocation of the SLC2A4/GLUT4 glucose transporter to the cell surface. Phosphorylation of PTPN1 at 'Ser-50' negatively modulates its phosphatase activity preventing dephosphorylation of the insulin receptor and the attenuation of insulin signaling. Phosphorylation of TBC1D4 triggers the binding of this effector to inhibitory 14-3-3 proteins, which is required for insulin-stimulated glucose transport. AKT regulates also the storage of glucose in the form of glycogen by phosphorylating GSK3A at 'Ser-21' and GSK3B at 'Ser-9', resulting in inhibition of its kinase activity. Phosphorylation of GSK3 isoforms by AKT is also thought to be one mechanism by which cell proliferation is driven. AKT regulates also cell survival via the phosphorylation of MAP3K5 (apoptosis signal-related kinase). Phosphorylation of 'Ser-83' decreases MAP3K5 kinase activity stimulated by oxidative stress and thereby prevents apoptosis. AKT mediates insulin-stimulated protein synthesis by phosphorylating TSC2 at 'Ser-939' and 'Thr-1462', thereby activating mTORC1 signaling and leading to both phosphorylation of 4E-BP1 and in activation of RPS6KB1. AKT is involved in the phosphorylation of members of the FOXO factors (Forkhead family of transcription factors), leading to binding of 14-3-3 proteins and cytoplasmic localization. In particular, FOXO1 is phosphorylated at 'Thr-24', 'Ser-256' and 'Ser-319'. FOXO3 and FOXO4 are phosphorylated on equivalent sites. AKT has an important role in the regulation of NF-kappa-B-dependent gene transcription and positively regulates the activity of CREB1 (cyclic AMP (cAMP)-response element binding protein). The phosphorylation of CREB1 induces the binding of accessory proteins that are necessary for the transcription of pro-survival genes such as BCL2 and MCL1. AKT phosphorylates 'Ser-454' on ATP citrate lyase (ACLY), thereby potentially regulating ACLY activity and fatty acid synthesis. Activates the 3B isoform of cyclic nucleotide phosphodiesterase (PDE3B) via phosphorylation of 'Ser-273', resulting in reduced cyclic AMP levels and inhibition of lipolysis. Phosphorylates PIKFYVE on 'Ser-318', which results in increased PI(3)P-5 activity. The Rho GTPase-activating protein DLC1 is another substrate and its phosphorylation is implicated in the regulation cell proliferation and cell growth. AKT plays a role as key modulator of the AKT-mTOR signaling pathway controlling the tempo of the process of newborn neurons integration during adult neurogenesis, including correct neuron positioning, dendritic development and synapse formation. Signals downstream of phosphatidylinositol 3-kinase (PI(3)K) to mediate the effects of various growth factors such as platelet-derived growth factor (PDGF), epidermal growth factor (EGF), insulin and insulin-like growth factor I (IGF-I). AKT mediates the antiapoptotic effects of IGF-I. Essential for the SPATA13-mediated regulation of cell migration and adhesion assembly and disassembly. May be involved in the regulation of the placental development. Involved in the inhibition of ciliogenesis associated with RAB8-dependent cilia growth (PubMed:31204173). {ECO:0000269|PubMed:31204173}.; FUNCTION: One of the few specific substrates of AKT2 identified recently is PITX2. Phosphorylation of PITX2 impairs its association with the CCND1 mRNA-stabilizing complex thus shortening the half-life of CCND1. AKT2 seems also to be the principal isoform responsible of the regulation of glucose uptake. Phosphorylates C2CD5 on 'Ser-197' during insulin-stimulated adipocytes. AKT2 is also specifically involved in skeletal muscle differentiation, one of its substrates in this process being ANKRD2. Down-regulation by RNA interference reduces the expression of the phosphorylated form of BAD, resulting in the induction of caspase-dependent apoptosis. Phosphorylates CLK2 on 'Thr-343'. |
 Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. |
 - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
 - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
Top |
Kinase-Substrate Information of AKT2_AKT2 |
 Phosphorylation target of the kinase Phosphorylation target of the kinase(phosphosite, 03-17-2024) |
| Kinase | Kinase UniProt Acc | Kinase species | Substrate | Substrate UniProt Acc | Substrate phosphorylated residues | Substrate phosphorylated sites (+/-7AA) | Domain |
| AKT2 | P31751 | human | ATP7A | Q04656 | S1424 | SYELPARsQIGQKsP | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S63 | ssQsstLsHsNRGsM | |
| AKT2 | P31751 | human | IRF3 | Q14653 | T180 | sPsLDNPtPFPNLGP | |
| AKT2 | P31751 | human | EGFR | P00533 | S229 | rCrGKSPsDCCHNQC | Furin-like |
| AKT2 | P31751 | human | PKP1 | Q13835 | T179 | LGsKGQKttQNRysF | |
| AKT2 | P31751 | human | EZR | P15311 | T567 | QGRDkyKtLRQIRQG | ERM_C |
| AKT2 | P31751 | human | CBY1 | Q9Y3M2 | S20 | TPPRKsAsLSNLHsL | Chibby |
| AKT2 | P31751 | human | HSF1 | Q00613 | S230 | PkYSRQFsLEHVHGS | |
| AKT2 | P31751 | human | BMI1 | P35226 | S316 | ANRPRKssVNGSSAT | |
| AKT2 | P31751 | human | ADAR | P55265 | T1033 | RLGERLRtMSCSDKI | A_deamin |
| AKT2 | P31751 | human | PKP1 | Q13835 | S191 | ysFystCsGQKAIKK | |
| AKT2 | P31751 | human | FLNC | Q14315 | S2233 | LGRERLGsFGsItRQ | Filamin |
| AKT2 | P31751 | human | PKP1 | Q13835 | T180 | GsKGQKttQNRysFy | |
| AKT2 | P31751 | human | IRS2 | Q9Y4H2 | S365 | sCRVRtAsEGDGGAA | |
| AKT2 | P31751 | human | H3C1 | P68431 | S10 | tkQtArkstGGkAPr | Histone |
| AKT2 | P31751 | human | WIPF1 | O43516 | T398 | TSRALPAtPQLPSrS | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S127 | ysQMENWsRHYPRGS | |
| AKT2 | P31751 | human | EDC3 | Q96F86 | S161 | sFRRRHNsWssssRH | Edc3_linker |
| AKT2 | P31751 | human | ATP7A | Q04656 | S1466 | ysRAsINsLLsDkRs | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S118 | EPDNRRFssysQMEN | |
| AKT2 | P31751 | human | PHB2 | Q99623 | S91 | RARPRkIssPTGSkD | Band_7 |
| AKT2 | P31751 | human | CHUK | O15111 | T23 | EMRERLGtGGFGNVC | Pkinase |
| AKT2 | P31751 | human | PHB2 | Q99623 | S176 | TERAKDFsLILDDVA | Band_7 |
| AKT2 | P31751 | human | H3C1 | P68431 | S28 | ATkAArksAPATGGV | Histone |
| AKT2 | P31751 | human | ADARB1 | P78563 | T553 | LQGERLLtMSCSDKI | A_deamin |
| AKT2 | P31751 | human | PKP1 | Q13835 | S65 | QsstLsHsNRGsMyD | |
| AKT2 | P31751 | human | LMNA | P02545 | S390 | EEErLRLsPsPtsQR | |
| AKT2 | P31751 | human | ATP7A | Q04656 | S1463 | IVNysRAsINsLLsD | |
| AKT2 | P31751 | human | PKP1 | Q13835 | T171 | RGtLRKGtLGsKGQK | |
| AKT2 | P31751 | human | PKP1 | Q13835 | T82 | ADNyNyGttsRssYy | |
| AKT2 | P31751 | human | IRF3 | Q14653 | S175 | PQPLRsPsLDNPtPF | |
| AKT2 | P31751 | human | PPIF | P30405 | S31 | LPAARACsKGsGDPS | |
| AKT2 | P31751 | human | PKP1 | Q13835 | T61 | sKssQsstLsHsNRG | |
| AKT2 | P31751 | human | GAPDH | P04406 | T237 | GMAFrVPtANVsVVD | Gp_dh_C |
| AKT2 | P31751 | human | PKP1 | Q13835 | S69 | LsHsNRGsMyDGLAD | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S174 | LRKGtLGsKGQKttQ | |
| AKT2 | P31751 | human | LMNA | P02545 | S404 | RsRGRAssHssQtQG | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S54 | MTVKRQKsKssQsst | |
| AKT2 | P31751 | human | MAP3K5 | Q99683 | S83 | ATRGRGssVGGGSrR | |
| AKT2 | P31751 | human | HTRA2 | O43464 | S212 | RVRVRLLsGDTYEAV | Trypsin_2; Peptidase_M50 |
| AKT2 | P31751 | human | PKP1 | Q13835 | S185 | KttQNRysFystCsG | |
| AKT2 | P31751 | human | XIAP | P98170 | S87 | VGRHRKVsPNCRFIN | BIR |
| AKT2 | P31751 | human | PKP1 | Q13835 | S57 | KRQKsKssQsstLsH | |
| AKT2 | P31751 | human | IRF3 | Q14653 | S14 | RILPWLVsQLDLGQL | IRF |
| AKT2 | P31751 | human | CYCS | P99999 | Y47 | ktGQAPGysytAANk | Cytochrom_C |
| AKT2 | P31751 | human | IRS2 | Q9Y4H2 | S1149 | GGRRRHssEtFSsTt | |
| AKT2 | P31751 | human | UPF1 | Q92900 | T151 | FCNGRGNtSGSHIVN | UPF1_Zn_bind |
| AKT2 | P31751 | human | GATA6 | Q92908-2 | S290 | KPQKRVPsSRRLGLS | |
| AKT2 | P31751 | human | ESR1 | P03372 | S167 | GGRERLAsTNDkGSM | Oest_recep |
| AKT2 | P31751 | human | PKP1 | Q13835 | T166 | LyCDPRGtLRKGtLG | |
| AKT2 | P31751 | human | PKP1 | Q13835 | T189 | NRysFystCsGQKAI | |
| AKT2 | P31751 | human | FSCN1 | Q16658 | T403 | GCRkVTGtLDANRSS | Fascin |
| AKT2 | P31751 | human | RACGAP1 | Q9H0H5 | T249 | WTRSRRktGtLQPWN | |
| AKT2 | P31751 | human | IRS2 | Q9Y4H2 | S306 | EFRPRsksQssGssA | |
| AKT2 | P31751 | human | RPS6KA3 | P51812 | T115 | KVRDRVRtKMERDIL | |
| AKT2 | P31751 | human | NF2 | P35240 | S10 | GAIASRMsFSsLkRK | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S188 | QNRysFystCsGQKA | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S84 | NyNyGttsRssYySK | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S60 | KsKssQsstLsHsNR | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S121 | NRRFssysQMENWsR | |
| AKT2 | P31751 | human | KHSRP | Q92945 | S193 | GLPERsVsLtGAPEs | KH_1 |
| AKT2 | P31751 | human | PIP5K1C | O60331 | S555 | RYRRRtQssGQDGRP | |
| AKT2 | P31751 | human | MAT1A | Q00266 | T202 | VIPVRIHtIVISVQH | S-AdoMet_synt_M |
| AKT2 | P31751 | human | HSF1 | Q00613 | S326 | ssVDtLLsPTALIDs | Vert_HS_TF |
| AKT2 | P31751 | human | CTNNB1 | P35222 | S552 | QDtQRRtsMGGtQQQ | |
| AKT2 | P31751 | human | ANKRD2 | Q9GZV1 | S99 | QERVRKTsLDLRREI | |
| AKT2 | P31751 | human | IRS2 | Q9Y4H2 | S577 | GLRKRtysLttPARQ | |
| AKT2 | P31751 | human | IRF3 | Q14653 | T207 | EEWEFEVtAFYRGRQ | IRF-3 |
| AKT2 | P31751 | human | C2CD5 | Q86YS7 | S197 | EARQRLIsLMsGELQ | |
| AKT2 | P31751 | human | SH3RF1 | Q7Z6J0 | S304 | KNTKKRHsFtsLtMA | |
| AKT2 | P31751 | human | PDK1 | Q15118 | T346 | APRPRVEtsRAVPLA | HATPase_c |
| AKT2 | P31751 | human | PKP1 | Q13835 | S59 | QKsKssQsstLsHsN | |
| AKT2 | P31751 | human | H3C1 | P68431 | T45 | PHryrPGtVALrEIR | Histone |
| AKT2 | P31751 | human | PKP1 | Q13835 | S119 | PDNRRFssysQMENW | |
| AKT2 | P31751 | human | EPRS1 | P07814 | S999 | NQGGGLsssGAGEGQ | |
| AKT2 | P31751 | human | WIPF1 | O43516 | S154 | PGrFPVPsPGHRSGP | |
| AKT2 | P31751 | human | PKP1 | Q13835 | S56 | VKRQKsKssQsstLs | |
| AKT2 | P31751 | human | MAT1A | Q00266 | S180 | LPWLRPDsKTQVTVQ | S-AdoMet_synt_M |
 Biological Network Integration of This Kinase and Substrates Biological Network Integration of This Kinase and Substrates (GeneMANIA website) |
 Enriched GO biological processes of the phosphorylation target genes of the kinase Enriched GO biological processes of the phosphorylation target genes of the kinase |
| Kinase | GOID | GO term | P.adjust |
| AKT2 | ID | Description | 0.00e+00 |
| AKT2 | GO:0034599 | cellular response to oxidative stress | 9.95e-05 |
| AKT2 | GO:0062197 | cellular response to chemical stress | 2.64e-04 |
| AKT2 | GO:0034614 | cellular response to reactive oxygen species | 4.30e-04 |
| AKT2 | GO:0071276 | cellular response to cadmium ion | 4.30e-04 |
| AKT2 | GO:0043281 | regulation of cysteine-type endopeptidase activity involved in apoptotic process | 4.30e-04 |
| AKT2 | GO:0006979 | response to oxidative stress | 5.07e-04 |
| AKT2 | GO:0052548 | regulation of endopeptidase activity | 5.85e-04 |
| AKT2 | GO:2000116 | regulation of cysteine-type endopeptidase activity | 7.62e-04 |
| AKT2 | GO:0052547 | regulation of peptidase activity | 7.73e-04 |
| AKT2 | GO:0000302 | response to reactive oxygen species | 8.13e-04 |
| AKT2 | GO:0046686 | response to cadmium ion | 1.24e-03 |
| AKT2 | GO:0008631 | intrinsic apoptotic signaling pathway in response to oxidative stress | 1.40e-03 |
| AKT2 | GO:0002221 | pattern recognition receptor signaling pathway | 1.40e-03 |
| AKT2 | GO:2001234 | negative regulation of apoptotic signaling pathway | 1.54e-03 |
| AKT2 | GO:0071456 | cellular response to hypoxia | 1.77e-03 |
| AKT2 | GO:0002758 | innate immune response-activating signaling pathway | 1.77e-03 |
| AKT2 | GO:0032481 | positive regulation of type I interferon production | 1.78e-03 |
| AKT2 | GO:0043122 | regulation of canonical NF-kappaB signal transduction | 1.95e-03 |
| AKT2 | GO:0036294 | cellular response to decreased oxygen levels | 1.99e-03 |
| AKT2 | GO:0002218 | activation of innate immune response | 2.19e-03 |
| AKT2 | GO:0051402 | neuron apoptotic process | 2.19e-03 |
| AKT2 | GO:0045088 | regulation of innate immune response | 2.35e-03 |
| AKT2 | GO:0071453 | cellular response to oxygen levels | 2.61e-03 |
| AKT2 | GO:0048732 | gland development | 2.61e-03 |
| AKT2 | GO:0001666 | response to hypoxia | 2.61e-03 |
| AKT2 | GO:0048145 | regulation of fibroblast proliferation | 2.61e-03 |
| AKT2 | GO:0007249 | canonical NF-kappaB signal transduction | 2.61e-03 |
| AKT2 | GO:0010464 | regulation of mesenchymal cell proliferation | 2.82e-03 |
| AKT2 | GO:0032872 | regulation of stress-activated MAPK cascade | 2.85e-03 |
| AKT2 | GO:0070302 | regulation of stress-activated protein kinase signaling cascade | 2.98e-03 |
| AKT2 | GO:0036293 | response to decreased oxygen levels | 2.99e-03 |
| AKT2 | GO:0097193 | intrinsic apoptotic signaling pathway | 3.10e-03 |
| AKT2 | GO:0002757 | immune response-activating signaling pathway | 3.10e-03 |
| AKT2 | GO:0043123 | positive regulation of canonical NF-kappaB signal transduction | 3.12e-03 |
| AKT2 | GO:0043086 | negative regulation of catalytic activity | 3.20e-03 |
| AKT2 | GO:0071248 | cellular response to metal ion | 3.24e-03 |
| AKT2 | GO:0010863 | positive regulation of phospholipase C activity | 3.32e-03 |
| AKT2 | GO:0071392 | cellular response to estradiol stimulus | 3.37e-03 |
| AKT2 | GO:0010586 | miRNA metabolic process | 3.37e-03 |
| AKT2 | GO:0048144 | fibroblast proliferation | 3.37e-03 |
| AKT2 | GO:1900274 | regulation of phospholipase C activity | 3.37e-03 |
| AKT2 | GO:0002764 | immune response-regulating signaling pathway | 3.37e-03 |
| AKT2 | GO:0045089 | positive regulation of innate immune response | 3.37e-03 |
| AKT2 | GO:0070482 | response to oxygen levels | 3.37e-03 |
| AKT2 | GO:0045216 | cell-cell junction organization | 3.57e-03 |
| AKT2 | GO:0032355 | response to estradiol | 3.87e-03 |
| AKT2 | GO:0010463 | mesenchymal cell proliferation | 4.42e-03 |
| AKT2 | GO:0071241 | cellular response to inorganic substance | 4.51e-03 |
| AKT2 | GO:0002833 | positive regulation of response to biotic stimulus | 4.51e-03 |
Top |
Related Drugs to AKT2_AKT2 |
 Drugs used for this fusion-positive patient. Drugs used for this fusion-positive patient. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Drug | Source | PMID |
 Distribution of the number of studies mentioning AKT2-AKT2 and kinase inhibitors the PubMed Abstract (04-01-2024) Distribution of the number of studies mentioning AKT2-AKT2 and kinase inhibitors the PubMed Abstract (04-01-2024) |
| Fusion gene - drug pair 1 | Fusion gene - drug pair 2 | PMID | Publication date | DOI | Study title |
Top |
Related Diseases to AKT2_AKT2 |
 Diseases that have this fusion gene. Diseases that have this fusion gene. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Disease | Source | PMID |
 Related diseases from the literature mentioned this fusion gene and drug. Related diseases from the literature mentioned this fusion gene and drug. (PubMed, 04-01-2024) |
| MeSH ID | MeSH term |
 Diseases associated with fusion partners. Diseases associated with fusion partners. (DisGeNet 4.0) |
| Partner | Gene | Disease ID | Disease name | # pubmeds | Source |
Top |
Clinical Trials of the Found Drugs/Small Molecules |
 Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) |
 Clinical Trials from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024) |
| Fusion Gene | Kinase Inhibitor | NCT ID | Study Status | Phases | Disease | # Enrolment | Date |

