| Description | potassium voltage-gated channel subfamily A member 2potassium channel, voltage gated shaker related subfamily A, member 2potassium voltage-gated channel, shaker-related subfamily, member 2voltage-gated K(+) channel HuKIVvoltage-gated potassium channel | serine/threonine-protein kinase greatwallgreatwall kinase homologgreatwall protein kinase |
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000369770, ENST00000316361,
ENST00000440270, ENST00000485317,
ENST00000525120, | ENST00000342386,
ENST00000375940, ENST00000375946,
ENST00000477034, |
| Hgene | Tgene |
KCNA2
P16389 | MASTL
Q96GX5 |
| FUNCTION: Voltage-gated potassium channel that mediates transmembrane potassium transport in excitable membranes, primarily in the brain and the central nervous system, but also in the cardiovascular system. Prevents aberrant action potential firing and regulates neuronal output. Forms tetrameric potassium-selective channels through which potassium ions pass in accordance with their electrochemical gradient. The channel alternates between opened and closed conformations in response to the voltage difference across the membrane (PubMed:19912772, PubMed:8495559, PubMed:11211111, PubMed:23769686). Can form functional homotetrameric channels and heterotetrameric channels that contain variable proportions of KCNA1, KCNA2, KCNA4, KCNA5, KCNA6, KCNA7, and possibly other family members as well; channel properties depend on the type of alpha subunits that are part of the channel (PubMed:8495559, PubMed:20220134). Channel properties are modulated by cytoplasmic beta subunits that regulate the subcellular location of the alpha subunits and promote rapid inactivation of delayed rectifier potassium channels. In vivo, membranes probably contain a mixture of heteromeric potassium channel complexes, making it difficult to assign currents observed in intact tissues to any particular potassium channel family member. Homotetrameric KCNA2 forms a delayed-rectifier potassium channel that opens in response to membrane depolarization, followed by slow spontaneous channel closure (PubMed:19912772, PubMed:23769686). In contrast, a heteromultimer formed by KCNA2 and KCNA4 shows rapid inactivation (PubMed:8495559). Regulates neuronal excitability and plays a role as pacemaker in the regulation of neuronal action potentials (By similarity). KCNA2-containing channels play a presynaptic role and prevent hyperexcitability and aberrant action potential firing (By similarity). Response to toxins that are selective for KCNA2-containing potassium channels suggests that in Purkinje cells, dendritic subthreshold KCNA2-containing potassium channels prevent random spontaneous calcium spikes, suppressing dendritic hyperexcitability without hindering the generation of somatic action potentials, and thereby play an important role in motor coordination (By similarity). Plays a role in the induction of long-term potentiation of neuron excitability in the CA3 layer of the hippocampus (By similarity). May function as down-stream effector for G protein-coupled receptors and inhibit GABAergic inputs to basolateral amygdala neurons (By similarity). May contribute to the regulation of neurotransmitter release, such as gamma-aminobutyric acid (GABA) (By similarity). Contributes to the regulation of the axonal release of the neurotransmitter dopamine (By similarity). Reduced KCNA2 expression plays a role in the perception of neuropathic pain after peripheral nerve injury, but not acute pain (By similarity). Plays a role in the regulation of the time spent in non-rapid eye movement (NREM) sleep (By similarity). {ECO:0000250|UniProtKB:P63141, ECO:0000250|UniProtKB:P63142, ECO:0000269|PubMed:11211111, ECO:0000269|PubMed:19912772, ECO:0000269|PubMed:20220134, ECO:0000269|PubMed:23769686, ECO:0000269|PubMed:8495559, ECO:0000305}. | FUNCTION: Serine/threonine kinase that plays a key role in M phase by acting as a regulator of mitosis entry and maintenance. Acts by promoting the inactivation of protein phosphatase 2A (PP2A) during M phase: does not directly inhibit PP2A but acts by mediating phosphorylation and subsequent activation of ARPP19 and ENSA at 'Ser-62' and 'Ser-67', respectively. ARPP19 and ENSA are phosphatase inhibitors that specifically inhibit the PPP2R2D (PR55-delta) subunit of PP2A. Inactivation of PP2A during M phase is essential to keep cyclin-B1-CDK1 activity high. Following DNA damage, it is also involved in checkpoint recovery by being inhibited. Phosphorylates histone protein in vitro; however such activity is unsure in vivo. May be involved in megakaryocyte differentiation. {ECO:0000269|PubMed:12890928, ECO:0000269|PubMed:19680222, ECO:0000269|PubMed:19793917, ECO:0000269|PubMed:20538976, ECO:0000269|PubMed:20818157}. |
| Kinase | GOID | GO term | P.adjust |
| MASTL | ID | Description | 0.00e+00 |
| MASTL | GO:0035308 | negative regulation of protein dephosphorylation | 9.69e-05 |
| MASTL | GO:0035305 | negative regulation of dephosphorylation | 9.69e-05 |
| MASTL | GO:0035304 | regulation of protein dephosphorylation | 3.18e-04 |
| MASTL | GO:0035303 | regulation of dephosphorylation | 4.53e-04 |
| MASTL | GO:0000086 | G2/M transition of mitotic cell cycle | 6.34e-04 |
| MASTL | GO:0044839 | cell cycle G2/M phase transition | 6.48e-04 |
| MASTL | GO:0006470 | protein dephosphorylation | 9.54e-04 |
| MASTL | GO:0016311 | dephosphorylation | 1.85e-03 |
| MASTL | GO:0045936 | negative regulation of phosphate metabolic process | 2.31e-03 |
| MASTL | GO:0010563 | negative regulation of phosphorus metabolic process | 2.31e-03 |
| MASTL | GO:0031400 | negative regulation of protein modification process | 2.82e-03 |
| MASTL | GO:0044772 | mitotic cell cycle phase transition | 2.99e-03 |
| MASTL | GO:0045722 | positive regulation of gluconeogenesis | 9.93e-03 |
| MASTL | GO:0046326 | positive regulation of glucose import | 1.62e-02 |
| MASTL | GO:0010828 | positive regulation of glucose transmembrane transport | 1.73e-02 |
| MASTL | GO:0010907 | positive regulation of glucose metabolic process | 1.73e-02 |
| MASTL | GO:0006111 | regulation of gluconeogenesis | 1.91e-02 |
| MASTL | GO:0046324 | regulation of glucose import | 2.05e-02 |
| MASTL | GO:0046323 | glucose import | 2.37e-02 |
| MASTL | GO:0010827 | regulation of glucose transmembrane transport | 2.37e-02 |
| MASTL | GO:0045913 | positive regulation of carbohydrate metabolic process | 2.37e-02 |
| MASTL | GO:0006094 | gluconeogenesis | 2.48e-02 |
| MASTL | GO:0019319 | hexose biosynthetic process | 2.48e-02 |
| MASTL | GO:0046364 | monosaccharide biosynthetic process | 2.48e-02 |
| MASTL | GO:0010906 | regulation of glucose metabolic process | 2.48e-02 |
| MASTL | GO:0043255 | regulation of carbohydrate biosynthetic process | 2.48e-02 |
| MASTL | GO:1904659 | glucose transmembrane transport | 2.60e-02 |
| MASTL | GO:0008645 | hexose transmembrane transport | 2.60e-02 |
| MASTL | GO:0015749 | monosaccharide transmembrane transport | 2.60e-02 |
| MASTL | GO:0034219 | carbohydrate transmembrane transport | 2.78e-02 |
| MASTL | GO:0062013 | positive regulation of small molecule metabolic process | 2.90e-02 |
| MASTL | GO:0007584 | response to nutrient | 2.95e-02 |
| MASTL | GO:0008643 | carbohydrate transport | 2.95e-02 |
| MASTL | GO:0050796 | regulation of insulin secretion | 2.97e-02 |
| MASTL | GO:0006109 | regulation of carbohydrate metabolic process | 3.06e-02 |
| MASTL | GO:0006006 | glucose metabolic process | 3.06e-02 |
| MASTL | GO:0090276 | regulation of peptide hormone secretion | 3.06e-02 |
| MASTL | GO:0002791 | regulation of peptide secretion | 3.06e-02 |
| MASTL | GO:0030073 | insulin secretion | 3.06e-02 |
| MASTL | GO:0090087 | regulation of peptide transport | 3.06e-02 |
| MASTL | GO:0016051 | carbohydrate biosynthetic process | 3.16e-02 |
| MASTL | GO:0034764 | positive regulation of transmembrane transport | 3.16e-02 |
| MASTL | GO:0019318 | hexose metabolic process | 3.31e-02 |
| MASTL | GO:0030072 | peptide hormone secretion | 3.31e-02 |
| MASTL | GO:0002790 | peptide secretion | 3.31e-02 |
| MASTL | GO:0046883 | regulation of hormone secretion | 3.31e-02 |
| MASTL | GO:0005996 | monosaccharide metabolic process | 3.31e-02 |
| MASTL | GO:0015833 | peptide transport | 3.31e-02 |
| MASTL | GO:0050708 | regulation of protein secretion | 3.31e-02 |

 Kinase Fusion gene summary
Kinase Fusion gene summary Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez
Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez  Kinase Fusion gene breakpoints across KCNA2 (5'-gene)
Kinase Fusion gene breakpoints across KCNA2 (5'-gene) Kinase Fusion gene breakpoints across MASTL (3'-gene)
Kinase Fusion gene breakpoints across MASTL (3'-gene) Kinase Fusion gene information.
Kinase Fusion gene information.  Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)
Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB.
Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB.  For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones.
For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. Four levels of functional features of fusion genes
Four levels of functional features of fusion genes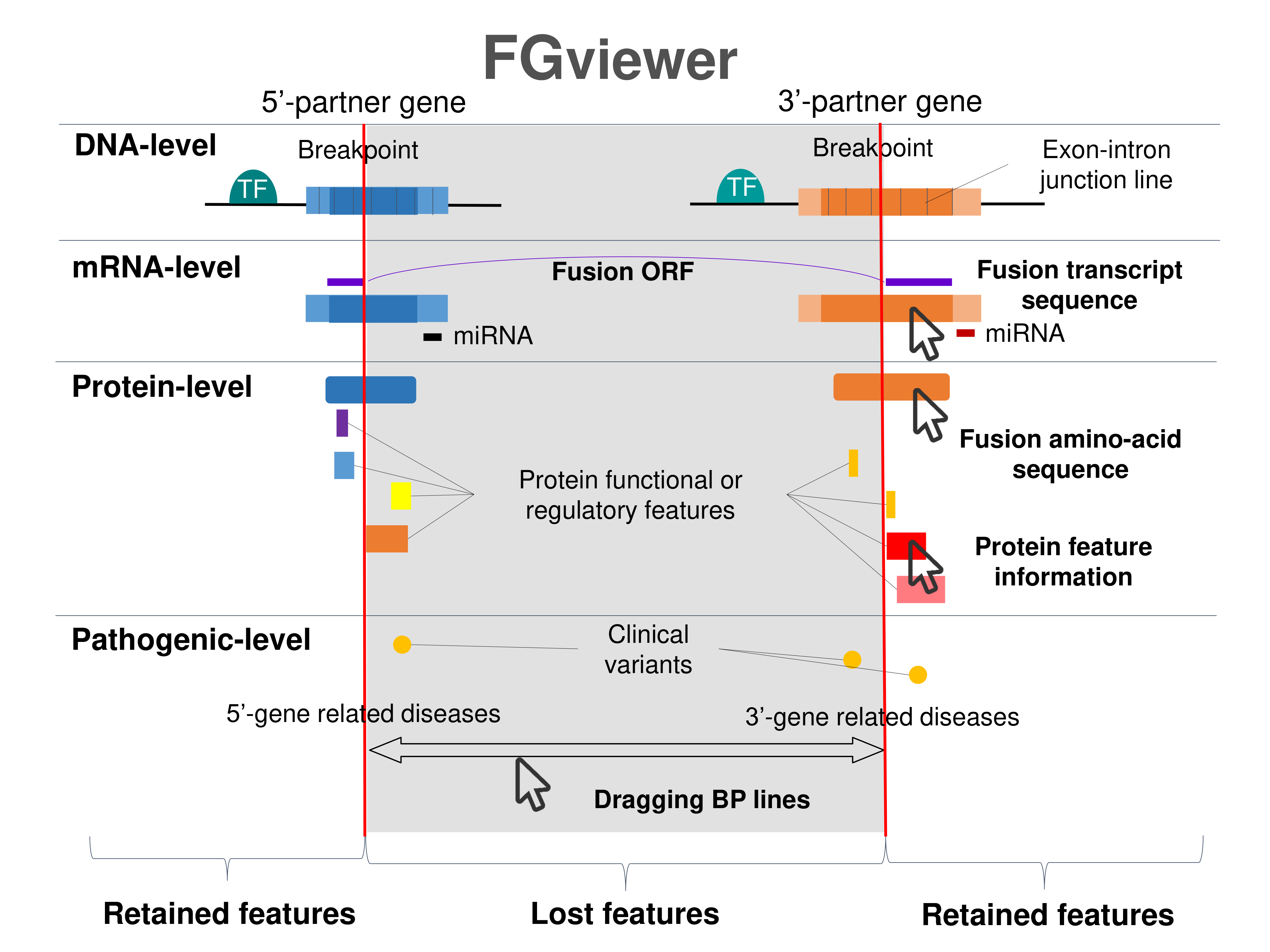
 Main function of each fusion partner protein. (from UniProt)
Main function of each fusion partner protein. (from UniProt) Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS.
Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt).
- Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt).
- Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). Phosphorylation target of the kinase
Phosphorylation target of the kinase Biological Network Integration of This Kinase and Substrates
Biological Network Integration of This Kinase and Substrates  Enriched GO biological processes of the phosphorylation target genes of the kinase
Enriched GO biological processes of the phosphorylation target genes of the kinase Drugs used for this fusion-positive patient.
Drugs used for this fusion-positive patient.  Distribution of the number of studies mentioning KCNA2-MASTL and kinase inhibitors the PubMed Abstract (04-01-2024)
Distribution of the number of studies mentioning KCNA2-MASTL and kinase inhibitors the PubMed Abstract (04-01-2024) Diseases that have this fusion gene.
Diseases that have this fusion gene.  Related diseases from the literature mentioned this fusion gene and drug.
Related diseases from the literature mentioned this fusion gene and drug.  Diseases associated with fusion partners.
Diseases associated with fusion partners.  Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024)
Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024)
Clinical Trials from clinicaltrials.gov (06-17-2024)
