| UTHEALTH HOME ABOUT SBMI A-Z WEBMAIL INSIDE THE UNIVERSITY |

|
|||||||
|
Kinase Fusion Gene:MARK3_EXOSC10 |
Kinase Fusion Protein Summary |
 Kinase Fusion gene summary Kinase Fusion gene summary |
| Kinase Fusion partner gene information | Kinase Fusion gene name: MARK3_EXOSC10 | KinaseFusionDB ID: KFG3645 | FusionGDB2.0 ID: KFG3645 | Hgene | Tgene | Gene symbol | MARK3 | EXOSC10 | Gene ID | 4140 | 5394 | |
| Gene name | microtubule affinity regulating kinase 3 | exosome component 10 | ||||||||||
| Synonyms | CTAK1|KP78|PAR1A|Par-1a|VIPB | PM-Scl|PM/Scl-100|PMSCL|PMSCL2|RRP6|Rrp6p|p2|p3|p4 | ||||||||||
| Cytomap | 14q32.32-q32.33 | 1p36.22 | ||||||||||
| Type of gene | protein-coding | protein-coding | ||||||||||
| Description | MAP/microtubule affinity-regulating kinase 3C-TAK1ELKL motif kinase 2EMK-2cdc25C-associated protein kinase 1protein kinase STK10ser/Thr protein kinase PAR-1serine/threonine-protein kinase p78 | exosome complex component 10exosome component 10P100 polymyositis-scleroderma overlap syndrome-associated autoantigenautoantigen PM-SCLpolymyositis/scleroderma autoantigen 100 kDapolymyositis/scleroderma autoantigen 2 | ||||||||||
| Modification date | 20240407 | 20240411 | ||||||||||
| UniProtAcc | P27448 | Q01780 | ||||||||||
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000216288, ENST00000303622, ENST00000335102, ENST00000416682, ENST00000429436, ENST00000440884, ENST00000553942, ENST00000561071, | ENST00000485606, ENST00000544779, ENST00000304457, ENST00000376936, | |||||||||
| Context (manual curation of fusion genes in KinaseFusionDB) | PubMed: MARK3 [Title/Abstract] AND EXOSC10 [Title/Abstract] AND fusion [Title/Abstract] | |||||||||||
| Most frequent breakpoint (based on all fusion genes of FusionGDB 2.0) | MARK3(103928798)-EXOSC10(11137708), # samples:1 | |||||||||||
 Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez |
| Partner | Gene | GO ID | GO term | PubMed ID |
| Hgene | MARK3 | GO:0018105 | peptidyl-serine phosphorylation | 9543386 |
| Hgene | MARK3 | GO:0032092 | positive regulation of protein binding | 9543386 |
| Hgene | MARK3 | GO:0035331 | negative regulation of hippo signaling | 28087714 |
| Hgene | MARK3 | GO:0036289 | peptidyl-serine autophosphorylation | 9543386 |
| Hgene | MARK3 | GO:1900181 | negative regulation of protein localization to nucleus | 16822840 |
| Tgene | EXOSC10 | GO:0006396 | RNA processing | 17174896|20531386 |
| Tgene | EXOSC10 | GO:0006401 | RNA catabolic process | 17174896|20531386 |
| Tgene | EXOSC10 | GO:0042274 | ribosomal small subunit biogenesis | 34516797 |
 Kinase Fusion gene breakpoints across MARK3 (5'-gene) Kinase Fusion gene breakpoints across MARK3 (5'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
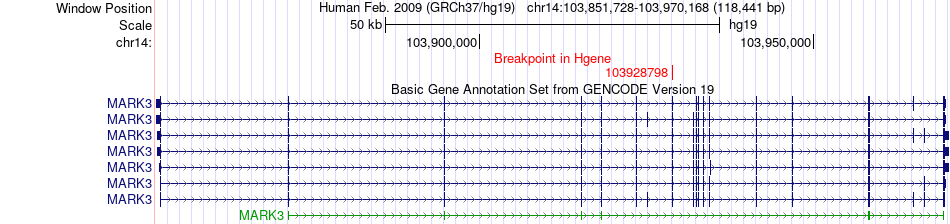 |
 Kinase Fusion gene breakpoints across EXOSC10 (3'-gene) Kinase Fusion gene breakpoints across EXOSC10 (3'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
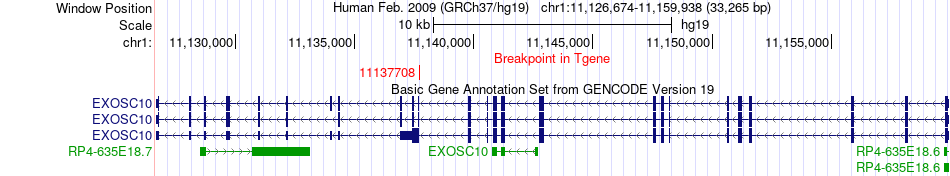 |
Top |
Kinase Fusion Gene Sample Information |
 Kinase Fusion gene information. Kinase Fusion gene information. |
 Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)* All genome coordinats were lifted-over on hg19. * Click on the break point to see the gene structure around the break point region using the UCSC Genome Browser. |
| Source | Sample | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp |
| ChimerDB4 | ERR315491 | MARK3 | chr14 | 103928798 | EXOSC10 | chr1 | 11137708 |
Top |
Kinase Fusion ORF Analysis |
 Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. |
| Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | Seq length (transcript) | Seq length (amino acids) |
Top |
Kinase Fusion Amino Acid Sequences |
 For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. |
| >Henst_Tenst_Hgene_Hchr_Hbp_Tgene_Tchr_Tbp_length(fusion AA)_AAseq |
Multiple Sequence Alignment of All Fusion Protein Isoforms |
Top |
Kinase Fusion Protein Functional Features |
 Four levels of functional features of fusion genes Four levels of functional features of fusion genesGo to FGviewer search page for the most frequent breakpoint (https://ccsmweb.uth.edu/FGviewer/chr14:103928798/chr1:11137708) - FGviewer provides the online visualization of the retention search of the protein functional features across DNA, RNA, protein, and pathological levels. - How to search 1. Put your fusion gene symbol. 2. Press the tab key until there will be shown the breakpoint information filled. 4. Go down and press 'Search' tab twice. 4. Go down to have the hyperlink of the search result. 5. Click the hyperlink. 6. See the FGviewer result for your fusion gene. |
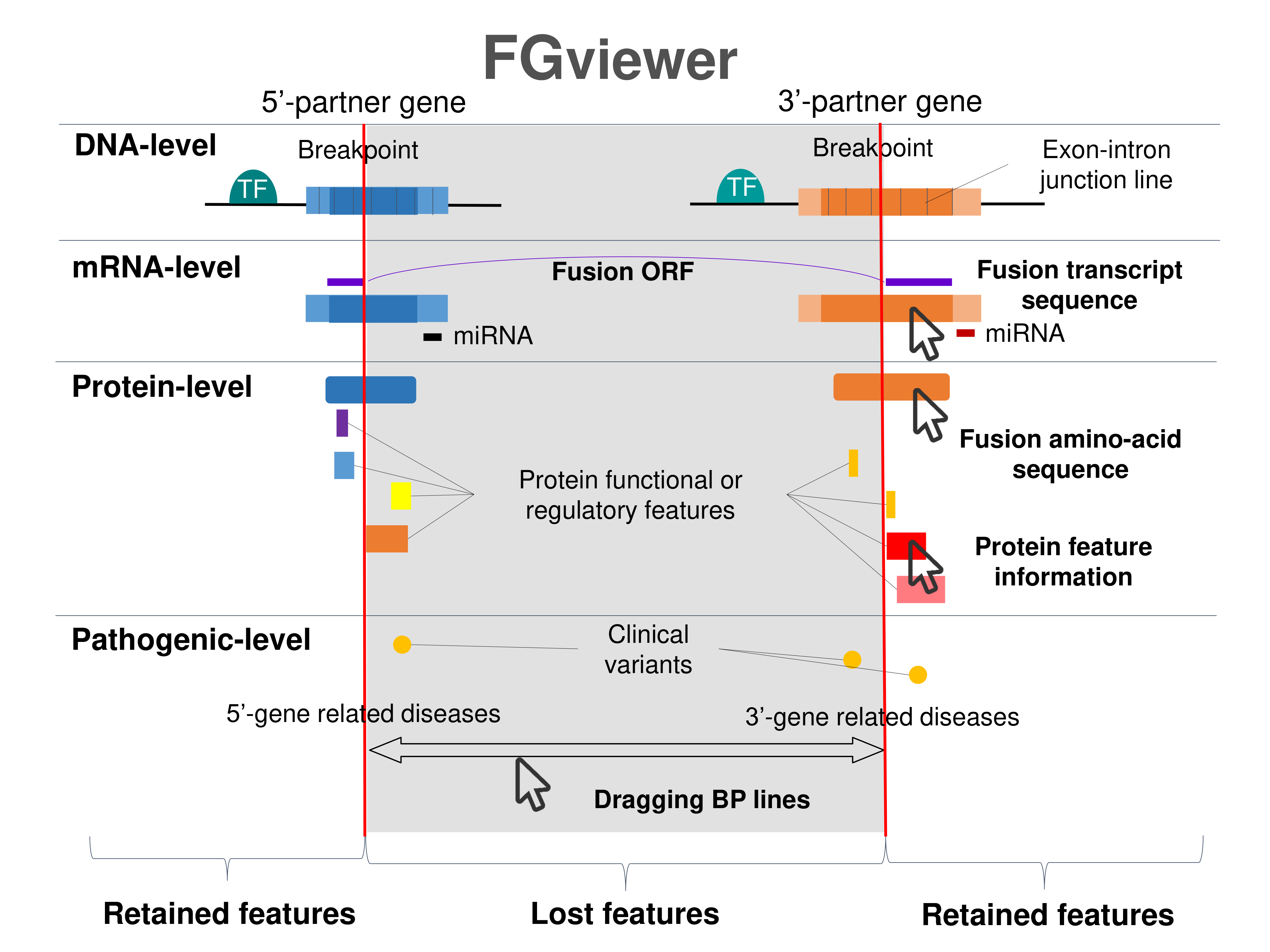 |
 Main function of each fusion partner protein. (from UniProt) Main function of each fusion partner protein. (from UniProt) |
| Hgene | Tgene |
| MARK3 | EXOSC10 |
| FUNCTION: Serine/threonine-protein kinase (PubMed:16822840, PubMed:16980613, PubMed:23666762). Involved in the specific phosphorylation of microtubule-associated proteins for MAP2 and MAP4. Phosphorylates the microtubule-associated protein MAPT/TAU (PubMed:23666762). Phosphorylates CDC25C on 'Ser-216' (PubMed:12941695). Regulates localization and activity of some histone deacetylases by mediating phosphorylation of HDAC7, promoting subsequent interaction between HDAC7 and 14-3-3 and export from the nucleus (PubMed:16980613). Regulates localization and activity of MITF by mediating its phosphorylation, promoting subsequent interaction between MITF and 14-3-3 and retention in the cytosol (PubMed:16822840). Negatively regulates the Hippo signaling pathway and antagonizes the phosphorylation of LATS1. Cooperates with DLG5 to inhibit the kinase activity of STK3/MST2 toward LATS1 (PubMed:28087714). Phosphorylates PKP2 and KSR1 (PubMed:12941695). {ECO:0000269|PubMed:12941695, ECO:0000269|PubMed:16822840, ECO:0000269|PubMed:16980613, ECO:0000269|PubMed:23666762, ECO:0000269|PubMed:28087714}. | FUNCTION: Catalytic component of the RNA exosome complex which has 3'->5' exoribonuclease activity and participates in a multitude of cellular RNA processing and degradation events. In the nucleus, the RNA exosome complex is involved in proper maturation of stable RNA species such as rRNA, snRNA and snoRNA, in the elimination of RNA processing by-products and non-coding 'pervasive' transcripts, such as antisense RNA species and promoter-upstream transcripts (PROMPTs), and of mRNAs with processing defects, thereby limiting or excluding their export to the cytoplasm. Part of the small subunit (SSU) processome, first precursor of the small eukaryotic ribosomal subunit. During the assembly of the SSU processome in the nucleolus, many ribosome biogenesis factors, an RNA chaperone and ribosomal proteins associate with the nascent pre-rRNA and work in concert to generate RNA folding, modifications, rearrangements and cleavage as well as targeted degradation of pre-ribosomal RNA by the RNA exosome (PubMed:34516797). The RNA exosome may be involved in Ig class switch recombination (CSR) and/or Ig variable region somatic hypermutation (SHM) by targeting AICDA deamination activity to transcribed dsDNA substrates. In the cytoplasm, the RNA exosome complex is involved in general mRNA turnover and specifically degrades inherently unstable mRNAs containing AU-rich elements (AREs) within their 3' untranslated regions, and in RNA surveillance pathways, preventing translation of aberrant mRNAs. It seems to be involved in degradation of histone mRNA. EXOSC10 is required for nucleolar localization of C1D and probably mediates the association of MTREX, C1D and MPHOSPH6 with the RNA exosome involved in the maturation of 5.8S rRNA. Plays a role in the recruitment of replication protein A complex (RPA) and RAD51 to DNA double-strand breaks caused by irradiation, contributing to DNA repair by homologous recombination (PubMed:31086179, PubMed:25632158). Regulates levels of damage-induced RNAs in order to prevent DNA-RNA hybrid formation at DNA double-strand breaks and limit DNA end resection after damage (PubMed:31086179). Plays a role in oocyte development, maturation and survival (By similarity). Required for normal testis development and mitotic division of spermatogonia (By similarity). Plays a role in proper embryo development (By similarity). Required for global protein translation (PubMed:36912080, PubMed:26857222). Required for cell proliferation (PubMed:36912080). Regulates metabolism of C9orf72-derived repeat RNA that can be translated into toxic dipeptide repeat proteins (PubMed:32830871). {ECO:0000250|UniProtKB:P56960, ECO:0000269|PubMed:14527413, ECO:0000269|PubMed:16455498, ECO:0000269|PubMed:17412707, ECO:0000269|PubMed:17545563, ECO:0000269|PubMed:18172165, ECO:0000269|PubMed:19056938, ECO:0000269|PubMed:20368444, ECO:0000269|PubMed:20699273, ECO:0000269|PubMed:25632158, ECO:0000269|PubMed:26857222, ECO:0000269|PubMed:31086179, ECO:0000269|PubMed:32830871, ECO:0000269|PubMed:34516797, ECO:0000269|PubMed:36912080}. |
 Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. |
 - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
 - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
Top |
Kinase-Substrate Information of MARK3_EXOSC10 |
 Phosphorylation target of the kinase Phosphorylation target of the kinase(phosphosite, 03-17-2024) |
| Kinase | Kinase UniProt Acc | Kinase species | Substrate | Substrate UniProt Acc | Substrate phosphorylated residues | Substrate phosphorylated sites (+/-7AA) | Domain |
| MARK3 | P27448-3 | human | MARK3 | P27448-3 | S601 | SQTRsRGstNLFsKL | |
| MARK3 | P27448-3 | human | MARK3 | P27448-3 | S598 | TPLSQTRsRGstNLF | |
| MARK3 | P27448-3 | human | MARK3 | P27448-3 | S606 | RGstNLFsKLTSKLT | |
| MARK3 | P27448-3 | human | MARK3 | P27448-3 | T602 | QTRsRGstNLFsKLT | |
| MARK3 | P27448 | human | MEF2C | Q06413 | S222 | GYGNPRNsPGLLVsP | |
| MARK3 | P27448 | human | PPP1R2 | P41236 | S72 | LMKIDEPstPyHSMM | IPP-2 |
| MARK3 | P27448 | human | CDC25C | P30307 | S216 | sGLyRsPsMPENLNR | M-inducer_phosp |
| MARK3 | P27448 | human | TNK1 | Q13470 | S502 | RMKGIsRsLEsVLsL | |
| MARK3 | P27448 | human | FEZ1 | Q99689 | S58 | SEIISFKsMEDLVNE | FEZ |
| MARK3 | P27448 | human | ARHGEF2 | Q92974 | S151 | LsLAksVsttNIAGH |
 Biological Network Integration of This Kinase and Substrates Biological Network Integration of This Kinase and Substrates (GeneMANIA website) |
 Enriched GO biological processes of the phosphorylation target genes of the kinase Enriched GO biological processes of the phosphorylation target genes of the kinase |
| Kinase | GOID | GO term | P.adjust |
Top |
Related Drugs to MARK3_EXOSC10 |
 Drugs used for this fusion-positive patient. Drugs used for this fusion-positive patient. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Drug | Source | PMID |
 Distribution of the number of studies mentioning MARK3-EXOSC10 and kinase inhibitors the PubMed Abstract (04-01-2024) Distribution of the number of studies mentioning MARK3-EXOSC10 and kinase inhibitors the PubMed Abstract (04-01-2024) |
| Fusion gene - drug pair 1 | Fusion gene - drug pair 2 | PMID | Publication date | DOI | Study title |
Top |
Related Diseases to MARK3_EXOSC10 |
 Diseases that have this fusion gene. Diseases that have this fusion gene. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Disease | Source | PMID |
 Related diseases from the literature mentioned this fusion gene and drug. Related diseases from the literature mentioned this fusion gene and drug. (PubMed, 04-01-2024) |
| MeSH ID | MeSH term |
 Diseases associated with fusion partners. Diseases associated with fusion partners. (DisGeNet 4.0) |
| Partner | Gene | Disease ID | Disease name | # pubmeds | Source |
Top |
Clinical Trials of the Found Drugs/Small Molecules |
 Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) |
 Clinical Trials from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024) |
| Fusion Gene | Kinase Inhibitor | NCT ID | Study Status | Phases | Disease | # Enrolment | Date |

