| UTHEALTH HOME ABOUT SBMI A-Z WEBMAIL INSIDE THE UNIVERSITY |

|
|||||||
|
Kinase Fusion Gene:MTOR_MTOR |
Kinase Fusion Protein Summary |
 Kinase Fusion gene summary Kinase Fusion gene summary |
| Kinase Fusion partner gene information | Kinase Fusion gene name: MTOR_MTOR | KinaseFusionDB ID: KFG3985 | FusionGDB2.0 ID: KFG3985 | Hgene | Tgene | Gene symbol | MTOR | MTOR | Gene ID | 2475 | 2475 | |
| Gene name | mechanistic target of rapamycin kinase | mechanistic target of rapamycin kinase | ||||||||||
| Synonyms | FRAP|FRAP1|FRAP2|RAFT1|RAPT1|SKS | FRAP|FRAP1|FRAP2|RAFT1|RAPT1|SKS | ||||||||||
| Cytomap | 1p36.22 | 1p36.22 | ||||||||||
| Type of gene | protein-coding | protein-coding | ||||||||||
| Description | serine/threonine-protein kinase mTORFK506 binding protein 12-rapamycin associated protein 2FK506-binding protein 12-rapamycin complex-associated protein 1FKBP-rapamycin associated proteinFKBP12-rapamycin complex-associated protein 1mammalian target o | serine/threonine-protein kinase mTORFK506 binding protein 12-rapamycin associated protein 2FK506-binding protein 12-rapamycin complex-associated protein 1FKBP-rapamycin associated proteinFKBP12-rapamycin complex-associated protein 1mammalian target o | ||||||||||
| Modification date | 20240411 | 20240411 | ||||||||||
| UniProtAcc | P42345 | P42345 | ||||||||||
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000361445, ENST00000376838, ENST00000495435, | ENST00000361445, ENST00000376838, ENST00000495435, | |||||||||
| Context (manual curation of fusion genes in KinaseFusionDB) | PubMed: MTOR [Title/Abstract] AND MTOR [Title/Abstract] AND fusion [Title/Abstract] | |||||||||||
| Most frequent breakpoint (based on all fusion genes of FusionGDB 2.0) | MTOR(11186756)-MTOR(11182129), # samples:1 | |||||||||||
 Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez |
| Partner | Gene | GO ID | GO term | PubMed ID |
| Hgene | MTOR | GO:0001558 | regulation of cell growth | 18762023 |
| Hgene | MTOR | GO:0006468 | protein phosphorylation | 15467718 |
| Hgene | MTOR | GO:0006468 | protein phosphorylation | 18925875 |
| Hgene | MTOR | GO:0007040 | lysosome organization | 22692423 |
| Hgene | MTOR | GO:0009267 | cellular response to starvation | 22343943|22576015|22692423|28223137 |
| Hgene | MTOR | GO:0010507 | negative regulation of autophagy | 22576015|30704899|32561715 |
| Hgene | MTOR | GO:0016242 | negative regulation of macroautophagy | 25327288 |
| Hgene | MTOR | GO:0016310 | phosphorylation | 25327288 |
| Hgene | MTOR | GO:0016310 | phosphorylation | 11853878 |
| Hgene | MTOR | GO:0018105 | peptidyl-serine phosphorylation | 22343943|22576015|22692423 |
| Hgene | MTOR | GO:0031648 | protein destabilization | 36608670 |
| Hgene | MTOR | GO:0031667 | response to nutrient levels | 29750193 |
| Hgene | MTOR | GO:0031669 | cellular response to nutrient levels | 29750193|32561715 |
| Hgene | MTOR | GO:0031670 | cellular response to nutrient | 22017875|22017876|22017877 |
| Hgene | MTOR | GO:0032869 | cellular response to insulin stimulus | 18372248 |
| Hgene | MTOR | GO:0034198 | cellular response to amino acid starvation | 22343943|22424946|22576015|22692423 |
| Hgene | MTOR | GO:0038202 | TORC1 signaling | 12087098|17517883|18372248|22017875|22017876|22017877|24403073|24448649|28223137|29236692|29750193|31112131|32612235|36608670 |
| Hgene | MTOR | GO:0043200 | response to amino acid | 18497260 |
| Hgene | MTOR | GO:0045727 | positive regulation of translation | 18762023 |
| Hgene | MTOR | GO:0045948 | positive regulation of translational initiation | 22578813|29750193 |
| Hgene | MTOR | GO:0046777 | protein autophosphorylation | 15467718 |
| Hgene | MTOR | GO:0051647 | nucleus localization | 22343943|22576015|22692423 |
| Hgene | MTOR | GO:0071230 | cellular response to amino acid stimulus | 22424946 |
| Hgene | MTOR | GO:0071233 | cellular response to L-leucine | 22424946 |
| Hgene | MTOR | GO:1900181 | negative regulation of protein localization to nucleus | 22692423|24448649|32612235|35662396 |
| Hgene | MTOR | GO:1905672 | negative regulation of lysosome organization | 24448649|32612235 |
| Hgene | MTOR | GO:1990253 | cellular response to leucine starvation | 22424946 |
| Hgene | MTOR | GO:2000785 | regulation of autophagosome assembly | 23524951 |
| Tgene | MTOR | GO:0001558 | regulation of cell growth | 18762023 |
| Tgene | MTOR | GO:0006468 | protein phosphorylation | 15467718 |
| Tgene | MTOR | GO:0006468 | protein phosphorylation | 18925875 |
| Tgene | MTOR | GO:0007040 | lysosome organization | 22692423 |
| Tgene | MTOR | GO:0009267 | cellular response to starvation | 22343943|22576015|22692423|28223137 |
| Tgene | MTOR | GO:0010507 | negative regulation of autophagy | 22576015|30704899|32561715 |
| Tgene | MTOR | GO:0016242 | negative regulation of macroautophagy | 25327288 |
| Tgene | MTOR | GO:0016310 | phosphorylation | 25327288 |
| Tgene | MTOR | GO:0016310 | phosphorylation | 11853878 |
| Tgene | MTOR | GO:0018105 | peptidyl-serine phosphorylation | 22343943|22576015|22692423 |
| Tgene | MTOR | GO:0031648 | protein destabilization | 36608670 |
| Tgene | MTOR | GO:0031667 | response to nutrient levels | 29750193 |
| Tgene | MTOR | GO:0031669 | cellular response to nutrient levels | 29750193|32561715 |
| Tgene | MTOR | GO:0031670 | cellular response to nutrient | 22017875|22017876|22017877 |
| Tgene | MTOR | GO:0032869 | cellular response to insulin stimulus | 18372248 |
| Tgene | MTOR | GO:0034198 | cellular response to amino acid starvation | 22343943|22424946|22576015|22692423 |
| Tgene | MTOR | GO:0038202 | TORC1 signaling | 12087098|17517883|18372248|22017875|22017876|22017877|24403073|24448649|28223137|29236692|29750193|31112131|32612235|36608670 |
| Tgene | MTOR | GO:0043200 | response to amino acid | 18497260 |
| Tgene | MTOR | GO:0045727 | positive regulation of translation | 18762023 |
| Tgene | MTOR | GO:0045948 | positive regulation of translational initiation | 22578813|29750193 |
| Tgene | MTOR | GO:0046777 | protein autophosphorylation | 15467718 |
| Tgene | MTOR | GO:0051647 | nucleus localization | 22343943|22576015|22692423 |
| Tgene | MTOR | GO:0071230 | cellular response to amino acid stimulus | 22424946 |
| Tgene | MTOR | GO:0071233 | cellular response to L-leucine | 22424946 |
| Tgene | MTOR | GO:1900181 | negative regulation of protein localization to nucleus | 22692423|24448649|32612235|35662396 |
| Tgene | MTOR | GO:1905672 | negative regulation of lysosome organization | 24448649|32612235 |
| Tgene | MTOR | GO:1990253 | cellular response to leucine starvation | 22424946 |
| Tgene | MTOR | GO:2000785 | regulation of autophagosome assembly | 23524951 |
 Kinase Fusion gene breakpoints across MTOR (5'-gene) Kinase Fusion gene breakpoints across MTOR (5'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 Kinase Fusion gene breakpoints across MTOR (3'-gene) Kinase Fusion gene breakpoints across MTOR (3'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
Top |
Kinase Fusion Gene Sample Information |
 Kinase Fusion gene information. Kinase Fusion gene information. |
 Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)* All genome coordinats were lifted-over on hg19. * Click on the break point to see the gene structure around the break point region using the UCSC Genome Browser. |
| Source | Sample | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp |
| ChiTaRS5.0 | BE830802 | MTOR | chr1 | 11186756 | MTOR | chr1 | 11182129 |
Top |
Kinase Fusion ORF Analysis |
 Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. |
| Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | Seq length (transcript) | Seq length (amino acids) |
Top |
Kinase Fusion Amino Acid Sequences |
 For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. |
| >Henst_Tenst_Hgene_Hchr_Hbp_Tgene_Tchr_Tbp_length(fusion AA)_AAseq |
Multiple Sequence Alignment of All Fusion Protein Isoforms |
Top |
Kinase Fusion Protein Functional Features |
 Four levels of functional features of fusion genes Four levels of functional features of fusion genesGo to FGviewer search page for the most frequent breakpoint (https://ccsmweb.uth.edu/FGviewer/:11186756/:11182129) - FGviewer provides the online visualization of the retention search of the protein functional features across DNA, RNA, protein, and pathological levels. - How to search 1. Put your fusion gene symbol. 2. Press the tab key until there will be shown the breakpoint information filled. 4. Go down and press 'Search' tab twice. 4. Go down to have the hyperlink of the search result. 5. Click the hyperlink. 6. See the FGviewer result for your fusion gene. |
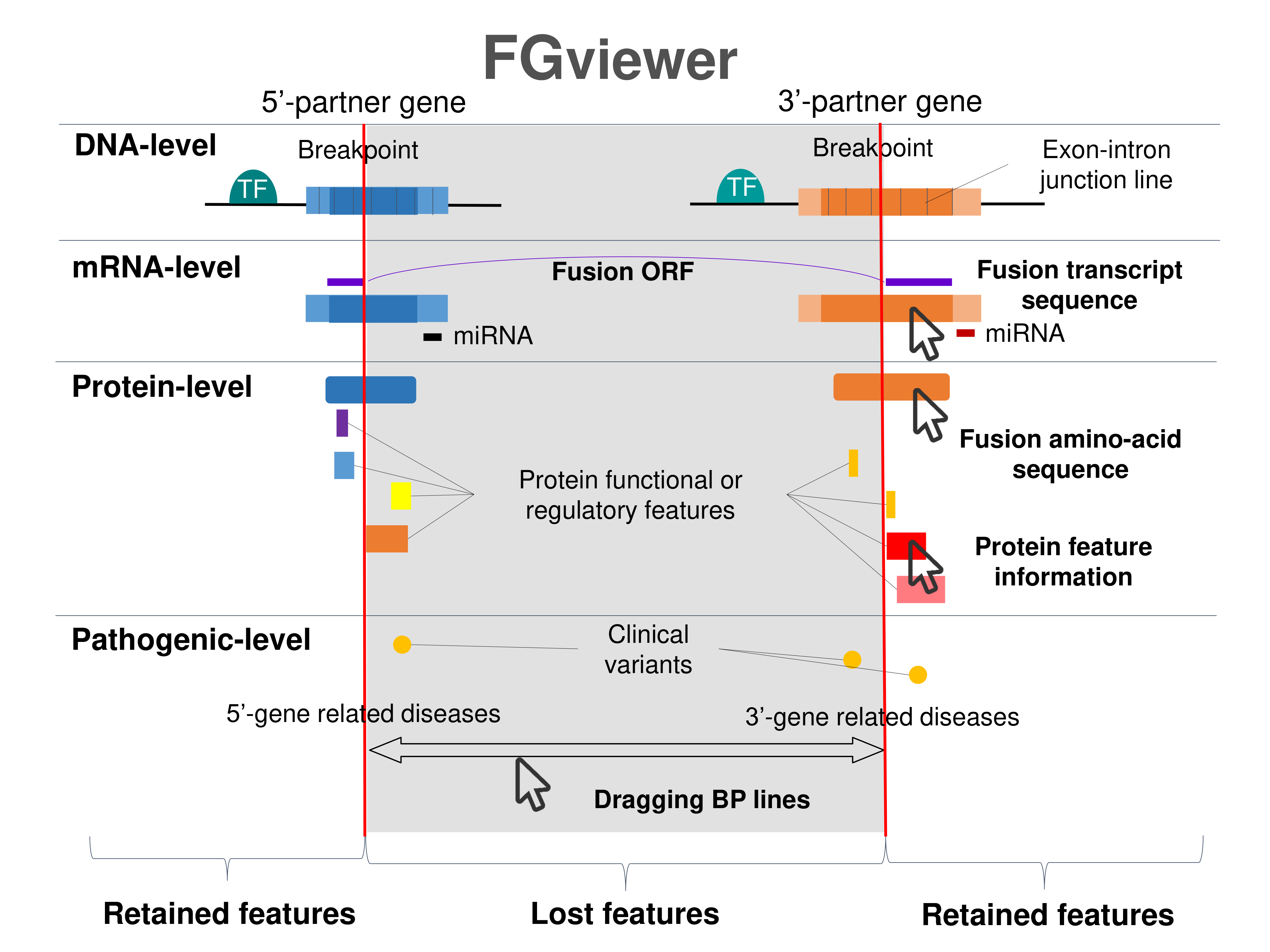 |
 Main function of each fusion partner protein. (from UniProt) Main function of each fusion partner protein. (from UniProt) |
| Hgene | Tgene |
| MTOR | MTOR |
| FUNCTION: Serine/threonine protein kinase which is a central regulator of cellular metabolism, growth and survival in response to hormones, growth factors, nutrients, energy and stress signals (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:31112131, PubMed:31601708, PubMed:32561715, PubMed:34519269, PubMed:29236692, PubMed:37751742). MTOR directly or indirectly regulates the phosphorylation of at least 800 proteins (PubMed:15268862, PubMed:15467718, PubMed:17517883, PubMed:18925875, PubMed:18372248, PubMed:18497260, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704, PubMed:29236692, PubMed:37751742). Functions as part of 2 structurally and functionally distinct signaling complexes mTORC1 and mTORC2 (mTOR complex 1 and 2) (PubMed:15268862, PubMed:15467718, PubMed:18925875, PubMed:18497260, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704). In response to nutrients, growth factors or amino acids, mTORC1 is recruited to the lysosome membrane and promotes protein, lipid and nucleotide synthesis by phosphorylating key regulators of mRNA translation and ribosome synthesis (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:31112131, PubMed:34519269, PubMed:29236692). This includes phosphorylation of EIF4EBP1 and release of its inhibition toward the elongation initiation factor 4E (eiF4E) (PubMed:24403073, PubMed:29236692). Moreover, phosphorylates and activates RPS6KB1 and RPS6KB2 that promote protein synthesis by modulating the activity of their downstream targets including ribosomal protein S6, eukaryotic translation initiation factor EIF4B, and the inhibitor of translation initiation PDCD4 (PubMed:12150925, PubMed:12087098, PubMed:18925875, PubMed:29150432, PubMed:29236692). Stimulates the pyrimidine biosynthesis pathway, both by acute regulation through RPS6KB1-mediated phosphorylation of the biosynthetic enzyme CAD, and delayed regulation, through transcriptional enhancement of the pentose phosphate pathway which produces 5-phosphoribosyl-1-pyrophosphate (PRPP), an allosteric activator of CAD at a later step in synthesis, this function is dependent on the mTORC1 complex (PubMed:23429704, PubMed:23429703). Regulates ribosome synthesis by activating RNA polymerase III-dependent transcription through phosphorylation and inhibition of MAF1 an RNA polymerase III-repressor (PubMed:20516213). Activates dormant ribosomes by mediating phosphorylation of SERBP1, leading to SERBP1 inactivation and reactivation of translation (PubMed:36691768). In parallel to protein synthesis, also regulates lipid synthesis through SREBF1/SREBP1 and LPIN1 (By similarity). To maintain energy homeostasis mTORC1 may also regulate mitochondrial biogenesis through regulation of PPARGC1A (By similarity). In the same time, mTORC1 inhibits catabolic pathways: negatively regulates autophagy through phosphorylation of ULK1 (PubMed:32561715). Under nutrient sufficiency, phosphorylates ULK1 at 'Ser-758', disrupting the interaction with AMPK and preventing activation of ULK1 (PubMed:32561715). Also prevents autophagy through phosphorylation of the autophagy inhibitor DAP (PubMed:20537536). Also prevents autophagy by phosphorylating RUBCNL/Pacer under nutrient-rich conditions (PubMed:30704899). Prevents autophagy by mediating phosphorylation of AMBRA1, thereby inhibiting AMBRA1 ability to mediate ubiquitination of ULK1 and interaction between AMBRA1 and PPP2CA (PubMed:23524951, PubMed:25438055). mTORC1 exerts a feedback control on upstream growth factor signaling that includes phosphorylation and activation of GRB10 a INSR-dependent signaling suppressor (PubMed:21659604). Among other potential targets mTORC1 may phosphorylate CLIP1 and regulate microtubules (PubMed:12231510). The mTORC1 complex is inhibited in response to starvation and amino acid depletion (PubMed:12150925, PubMed:12150926, PubMed:24403073). The non-canonical mTORC1 complex, which acts independently of RHEB, specifically mediates phosphorylation of MiT/TFE factors MITF, TFEB and TFE3 in the presence of nutrients, promoting their cytosolic retention and inactivation (PubMed:22576015, PubMed:22343943, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670, PubMed:36697823). Upon starvation or lysosomal stress, inhibition of mTORC1 induces dephosphorylation and nuclear translocation of TFEB and TFE3, promoting their transcription factor activity (PubMed:22576015, PubMed:22343943, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670). The mTORC1 complex regulates pyroptosis in macrophages by promoting GSDMD oligomerization (PubMed:34289345). MTOR phosphorylates RPTOR which in turn inhibits mTORC1 (By similarity). As part of the mTORC2 complex MTOR may regulate other cellular processes including survival and organization of the cytoskeleton (PubMed:15268862, PubMed:15467718). mTORC2 plays a critical role in the phosphorylation at 'Ser-473' of AKT1, a pro-survival effector of phosphoinositide 3-kinase, facilitating its activation by PDK1 (PubMed:15718470). mTORC2 may regulate the actin cytoskeleton, through phosphorylation of PRKCA, PXN and activation of the Rho-type guanine nucleotide exchange factors RHOA and RAC1A or RAC1B (PubMed:15268862). mTORC2 also regulates the phosphorylation of SGK1 at 'Ser-422' (PubMed:18925875). Regulates osteoclastogenesis by adjusting the expression of CEBPB isoforms (By similarity). Plays an important regulatory role in the circadian clock function; regulates period length and rhythm amplitude of the suprachiasmatic nucleus (SCN) and liver clocks (By similarity). Phosphorylates SQSTM1, promoting interaction between SQSTM1 and KEAP1 and subsequent inactivation of the BCR(KEAP1) complex (By similarity). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:12087098, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12150926, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:12718876, ECO:0000269|PubMed:14651849, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15545625, ECO:0000269|PubMed:15718470, ECO:0000269|PubMed:17517883, ECO:0000269|PubMed:18372248, ECO:0000269|PubMed:18497260, ECO:0000269|PubMed:18762023, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:20516213, ECO:0000269|PubMed:20537536, ECO:0000269|PubMed:21576368, ECO:0000269|PubMed:21659604, ECO:0000269|PubMed:22343943, ECO:0000269|PubMed:22576015, ECO:0000269|PubMed:22692423, ECO:0000269|PubMed:23429703, ECO:0000269|PubMed:23429704, ECO:0000269|PubMed:23524951, ECO:0000269|PubMed:24403073, ECO:0000269|PubMed:24448649, ECO:0000269|PubMed:25438055, ECO:0000269|PubMed:25799227, ECO:0000269|PubMed:26018084, ECO:0000269|PubMed:29150432, ECO:0000269|PubMed:29236692, ECO:0000269|PubMed:30704899, ECO:0000269|PubMed:31112131, ECO:0000269|PubMed:31601708, ECO:0000269|PubMed:32561715, ECO:0000269|PubMed:32612235, ECO:0000269|PubMed:34289345, ECO:0000269|PubMed:34519269, ECO:0000269|PubMed:36608670, ECO:0000269|PubMed:36691768, ECO:0000269|PubMed:36697823, ECO:0000269|PubMed:37751742}. | FUNCTION: Serine/threonine protein kinase which is a central regulator of cellular metabolism, growth and survival in response to hormones, growth factors, nutrients, energy and stress signals (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:31112131, PubMed:31601708, PubMed:32561715, PubMed:34519269, PubMed:29236692, PubMed:37751742). MTOR directly or indirectly regulates the phosphorylation of at least 800 proteins (PubMed:15268862, PubMed:15467718, PubMed:17517883, PubMed:18925875, PubMed:18372248, PubMed:18497260, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704, PubMed:29236692, PubMed:37751742). Functions as part of 2 structurally and functionally distinct signaling complexes mTORC1 and mTORC2 (mTOR complex 1 and 2) (PubMed:15268862, PubMed:15467718, PubMed:18925875, PubMed:18497260, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704). In response to nutrients, growth factors or amino acids, mTORC1 is recruited to the lysosome membrane and promotes protein, lipid and nucleotide synthesis by phosphorylating key regulators of mRNA translation and ribosome synthesis (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:31112131, PubMed:34519269, PubMed:29236692). This includes phosphorylation of EIF4EBP1 and release of its inhibition toward the elongation initiation factor 4E (eiF4E) (PubMed:24403073, PubMed:29236692). Moreover, phosphorylates and activates RPS6KB1 and RPS6KB2 that promote protein synthesis by modulating the activity of their downstream targets including ribosomal protein S6, eukaryotic translation initiation factor EIF4B, and the inhibitor of translation initiation PDCD4 (PubMed:12150925, PubMed:12087098, PubMed:18925875, PubMed:29150432, PubMed:29236692). Stimulates the pyrimidine biosynthesis pathway, both by acute regulation through RPS6KB1-mediated phosphorylation of the biosynthetic enzyme CAD, and delayed regulation, through transcriptional enhancement of the pentose phosphate pathway which produces 5-phosphoribosyl-1-pyrophosphate (PRPP), an allosteric activator of CAD at a later step in synthesis, this function is dependent on the mTORC1 complex (PubMed:23429704, PubMed:23429703). Regulates ribosome synthesis by activating RNA polymerase III-dependent transcription through phosphorylation and inhibition of MAF1 an RNA polymerase III-repressor (PubMed:20516213). Activates dormant ribosomes by mediating phosphorylation of SERBP1, leading to SERBP1 inactivation and reactivation of translation (PubMed:36691768). In parallel to protein synthesis, also regulates lipid synthesis through SREBF1/SREBP1 and LPIN1 (By similarity). To maintain energy homeostasis mTORC1 may also regulate mitochondrial biogenesis through regulation of PPARGC1A (By similarity). In the same time, mTORC1 inhibits catabolic pathways: negatively regulates autophagy through phosphorylation of ULK1 (PubMed:32561715). Under nutrient sufficiency, phosphorylates ULK1 at 'Ser-758', disrupting the interaction with AMPK and preventing activation of ULK1 (PubMed:32561715). Also prevents autophagy through phosphorylation of the autophagy inhibitor DAP (PubMed:20537536). Also prevents autophagy by phosphorylating RUBCNL/Pacer under nutrient-rich conditions (PubMed:30704899). Prevents autophagy by mediating phosphorylation of AMBRA1, thereby inhibiting AMBRA1 ability to mediate ubiquitination of ULK1 and interaction between AMBRA1 and PPP2CA (PubMed:23524951, PubMed:25438055). mTORC1 exerts a feedback control on upstream growth factor signaling that includes phosphorylation and activation of GRB10 a INSR-dependent signaling suppressor (PubMed:21659604). Among other potential targets mTORC1 may phosphorylate CLIP1 and regulate microtubules (PubMed:12231510). The mTORC1 complex is inhibited in response to starvation and amino acid depletion (PubMed:12150925, PubMed:12150926, PubMed:24403073). The non-canonical mTORC1 complex, which acts independently of RHEB, specifically mediates phosphorylation of MiT/TFE factors MITF, TFEB and TFE3 in the presence of nutrients, promoting their cytosolic retention and inactivation (PubMed:22576015, PubMed:22343943, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670, PubMed:36697823). Upon starvation or lysosomal stress, inhibition of mTORC1 induces dephosphorylation and nuclear translocation of TFEB and TFE3, promoting their transcription factor activity (PubMed:22576015, PubMed:22343943, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670). The mTORC1 complex regulates pyroptosis in macrophages by promoting GSDMD oligomerization (PubMed:34289345). MTOR phosphorylates RPTOR which in turn inhibits mTORC1 (By similarity). As part of the mTORC2 complex MTOR may regulate other cellular processes including survival and organization of the cytoskeleton (PubMed:15268862, PubMed:15467718). mTORC2 plays a critical role in the phosphorylation at 'Ser-473' of AKT1, a pro-survival effector of phosphoinositide 3-kinase, facilitating its activation by PDK1 (PubMed:15718470). mTORC2 may regulate the actin cytoskeleton, through phosphorylation of PRKCA, PXN and activation of the Rho-type guanine nucleotide exchange factors RHOA and RAC1A or RAC1B (PubMed:15268862). mTORC2 also regulates the phosphorylation of SGK1 at 'Ser-422' (PubMed:18925875). Regulates osteoclastogenesis by adjusting the expression of CEBPB isoforms (By similarity). Plays an important regulatory role in the circadian clock function; regulates period length and rhythm amplitude of the suprachiasmatic nucleus (SCN) and liver clocks (By similarity). Phosphorylates SQSTM1, promoting interaction between SQSTM1 and KEAP1 and subsequent inactivation of the BCR(KEAP1) complex (By similarity). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:12087098, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12150926, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:12718876, ECO:0000269|PubMed:14651849, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15545625, ECO:0000269|PubMed:15718470, ECO:0000269|PubMed:17517883, ECO:0000269|PubMed:18372248, ECO:0000269|PubMed:18497260, ECO:0000269|PubMed:18762023, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:20516213, ECO:0000269|PubMed:20537536, ECO:0000269|PubMed:21576368, ECO:0000269|PubMed:21659604, ECO:0000269|PubMed:22343943, ECO:0000269|PubMed:22576015, ECO:0000269|PubMed:22692423, ECO:0000269|PubMed:23429703, ECO:0000269|PubMed:23429704, ECO:0000269|PubMed:23524951, ECO:0000269|PubMed:24403073, ECO:0000269|PubMed:24448649, ECO:0000269|PubMed:25438055, ECO:0000269|PubMed:25799227, ECO:0000269|PubMed:26018084, ECO:0000269|PubMed:29150432, ECO:0000269|PubMed:29236692, ECO:0000269|PubMed:30704899, ECO:0000269|PubMed:31112131, ECO:0000269|PubMed:31601708, ECO:0000269|PubMed:32561715, ECO:0000269|PubMed:32612235, ECO:0000269|PubMed:34289345, ECO:0000269|PubMed:34519269, ECO:0000269|PubMed:36608670, ECO:0000269|PubMed:36691768, ECO:0000269|PubMed:36697823, ECO:0000269|PubMed:37751742}. |
 Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. |
 - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
 - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
Top |
Kinase-Substrate Information of MTOR_MTOR |
 Phosphorylation target of the kinase Phosphorylation target of the kinase(phosphosite, 03-17-2024) |
| Kinase | Kinase UniProt Acc | Kinase species | Substrate | Substrate UniProt Acc | Substrate phosphorylated residues | Substrate phosphorylated sites (+/-7AA) | Domain |
| MTOR | P42345 | human | ANKRD17 | O75179 | S2047 | VssPssPsPPAQPGG | |
| MTOR | P42345 | human | IRS1 | P35568 | S636 | sGDyMPMsPKsVSAP | |
| MTOR | P42345 | human | RRAGC | Q9HB90 | S21 | GsYGAADsFPKDFGY | |
| MTOR | P42345 | human | MKNK2 | Q9HBH9 | S74 | KRGRAtDsFsGRFED | |
| MTOR | P42345 | human | ULK1 | O75385 | S758 | PVVFtVGsPPsGStP | |
| MTOR | P42345 | human | BRD9 | Q9H8M2 | S588 | DPYEFLQsPEPAAsA | |
| MTOR | P42345 | human | WASHC2A | Q641Q2 | S700 | DDVDSGGsLFGsPPT | |
| MTOR | P42345 | human | ESR1 | P03372 | S104 | FPPLNsVsPsPLMLL | Oest_recep |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S549 | RKItsLSssLDTsLD | |
| MTOR | P42345 | human | TFEB | P19484 | S122 | PkPPPAAsPGVRAGH | MITF_TFEB_C_3_N |
| MTOR | P42345 | human | SRRM1 | Q8IYB3 | T574 | PRRRRtPtPPPRRRt | |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | S143 | LLYsKstsLtFHWKL | |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | T145 | YsKstsLtFHWKLWG | |
| MTOR | P42345 | human | MTOR | P42345 | S2448 | RsRtRtDsysAGQsV | |
| MTOR | P42345 | human | DAP | P51397 | S51 | DQEWEsPsPPkPtVF | DAP |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | S25 | PGIPPAyssPRRERL | |
| MTOR | P42345 | human | PKN2 | Q16513 | T958 | TsEAPILtPPREPRI | Pkinase_C |
| MTOR | P42345 | human | RPS6KB1 | P23443 | T412 | NQVFLGFtyVAPsVL | Pkinase_C |
| MTOR | P42345 | human | MCL1 | Q07820 | S64 | IGGSAGAsPPStLtP | |
| MTOR | P42345 | human | AKT1 | P31749 | S473 | RPHFPQFsysAsGtA | Pkinase_C |
| MTOR | P42345 | human | NRBF2 | Q96F24 | S120 | sPLsQkYsPSTEKCL | NRBF2 |
| MTOR | P42345 | human | SRRM1 | Q8IYB3 | T572 | PPPRRRRtPtPPPRR | |
| MTOR | P42345 | human | ELP1 | O95163 | S1174 | SETSsVVsGSEMSGK | |
| MTOR | P42345 | human | NAA10 | P41227 | S228 | StDVKDssEAsDSAS | |
| MTOR | P42345 | human | TFEB | P19484 | S211 | LVGVTSSsCPADLTQ | |
| MTOR | P42345 | human | PRKN | O60260 | S127 | AVILHTDsRkDsPPA | |
| MTOR | P42345 | human | AR | P10275 | S96 | QQQGEDGsPQAHRRG | Androgen_recep |
| MTOR | P42345 | human | MTOR | P42345 | S2478 | tGttVPEsIHsFIGD | |
| MTOR | P42345 | human | PASK | Q96RG2 | T640 | MAGLSFGtPtLDEPW | |
| MTOR | P42345 | human | AKT1S1 | Q96B36 | S183 | PTQQYAKsLPVSVPV | PRAS |
| MTOR | P42345 | human | LARP6 | Q9BRS8 | S409 | GRLNCStsPEIFRKC | |
| MTOR | P42345 | human | SRRM2 | Q9UQ35 | S1318 | sPEHKELsNsPLREN | |
| MTOR | P42345 | human | UNK | Q9C0B0 | S611 | GtsAsHGsLGLNGMN | |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S498 | GFsGGIPsPDKGHRK | |
| MTOR | P42345 | human | DNMT1 | P26358 | S714 | DNIPEMPsPkKMHQG | |
| MTOR | P42345 | human | ULK1 | O75385 | S638 | FDFPKtPssQNLLAL | |
| MTOR | P42345 | human | MAPKAP1 | Q9BPZ7 | S260 | PIHKFGFsTLALVEK | CRIM |
| MTOR | P42345 | human | UNK | Q9C0B0 | S606 | ENTFLGtsAsHGsLG | |
| MTOR | P42345 | human | HOXB13 | Q92826 | S31 | GRNLVAHsPLTSHPA | |
| MTOR | P42345 | human | AKT1 | P31749 | T450 | tAQMItItPPDQDDs | Pkinase_C |
| MTOR | P42345 | human | RRM1 | P23921 | S631 | IYTRRVLsGEFQIVN | Ribonuc_red_lgC |
| MTOR | P42345 | human | GRB10 | Q13322 | S476 | MNILGsQsPLHPSTL | |
| MTOR | P42345 | human | DUSP10 | Q9Y6W6 | S224 | SCREGKDsFKRIFsK | Rhodanese |
| MTOR | P42345 | human | WASHC2A | Q641Q2 | S704 | SGGsLFGsPPTSVPP | |
| MTOR | P42345 | human | PRKCE | Q02156 | T710 | TREEPVLtLVDEAIV | Pkinase_C |
| MTOR | P42345 | human | STK11IP | Q8N1F8 | S404 | EPRTLNPsPAGWFVQ | |
| MTOR | P42345 | human | LARP1 | Q6PKG0 | S774 | LPttVPEsPNyRNtR | |
| MTOR | P42345 | human | UNK | Q9C0B0 | S336 | QPSsAVssPtQPGPV | |
| MTOR | P42345 | human | UNK | Q9C0B0 | S608 | TFLGtsAsHGsLGLN | |
| MTOR | P42345 | human | MAF1 | Q9H063 | S60 | PHVLEALsPPQtsGL | Maf1 |
| MTOR | P42345 | human | AMOTL2 | Q9Y2J4 | S759 | SsSQRAAsLDsVATS | |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | S44 | tPGGtLFsttPGGtR | eIF_4EBP |
| MTOR | P42345 | human | PASK | Q96RG2 | S953 | FLAsLPGstHsTAAE | |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S582 | GHANVHPsQEQGEAL | |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | S141 | RMLLYsKstsLtFHW | |
| MTOR | P42345 | human | MTOR | P42345 | T2473 | PAHKKtGttVPEsIH | |
| MTOR | P42345 | human | MYCN | P04198 | S62 | LLPtPPLsPsrGFAE | Myc_N |
| MTOR | P42345 | human | RPTOR | Q8N122 | S859 | DtssLtQsAPAsPtN | |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | S65 | FLMECrNsPVtktPP | eIF_4EBP |
| MTOR | P42345 | human | NDRG1 | Q92597 | T346 | GtRsRsHtsEGtRsR | |
| MTOR | P42345 | human | SOD1 | P00441 | T40 | WGsIkGLtEGLHGFH | Sod_Cu |
| MTOR | P42345 | human | PRKCB | P05771 | T642 | TRQPVELtPtDKLFI | Pkinase_C |
| MTOR | P42345 | human | EEF2K | O00418 | S72 | ksERysssGsPANsF | |
| MTOR | P42345 | human | SRRM2 | Q9UQ35 | S1329 | LRENsFGsPLEFRNs | |
| MTOR | P42345 | human | MTOR | P42345 | S2454 | DsysAGQsVEILDGV | |
| MTOR | P42345 | human | MTOR | P42345 | S2481 | tVPEsIHsFIGDGLV | |
| MTOR | P42345 | human | ELL | P55199 | S309 | LGDPAAssPPGERGR | |
| MTOR | P42345 | human | EEF2K | O00418 | S74 | ERysssGsPANsFHF | |
| MTOR | P42345 | human | ESR1 | P03372 | S118 | LHPPPQLsPFLQPHG | Oest_recep |
| MTOR | P42345 | human | DEPTOR | Q8TB45 | T295 | GyFsssPtLsssPPV | |
| MTOR | P42345 | human | RPS6KB1 | P23443 | S394 | TRQtPVDsPDDStLS | Pkinase_C |
| MTOR | P42345 | human | PASK | Q96RG2 | S956 | sLPGstHsTAAELTG | |
| MTOR | P42345 | human | ESR1 | P03372 | S106 | PLNsVsPsPLMLLHP | Oest_recep |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | S26 | GIPPAyssPRRERLR | |
| MTOR | P42345 | human | DEPTOR | Q8TB45 | S293 | ssGyFsssPtLsssP | |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | S139 | AFRMLLYsKstsLtF | |
| MTOR | P42345 | human | HOXB13 | Q92826 | T41 | TSHPAAPtLMPAVNY | |
| MTOR | P42345 | human | BAG3 | O95817 | T285 | GsPARsstPLHsPsP | |
| MTOR | P42345 | human | RPRD1B | Q9NQG5 | S166 | DDyPGsysPQDPsAG | |
| MTOR | P42345 | human | SLC7A11 | Q9UPY5 | S26 | NVNGRLPsLGNkEPP | |
| MTOR | P42345 | human | PASK | Q96RG2 | T642 | GLSFGtPtLDEPWLG | |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | T70 | rNsPVtktPPRDLPt | eIF_4EBP |
| MTOR | P42345 | human | PRKCE | Q02156 | S729 | QEEFKGFsYFGEDLM | Pkinase_C |
| MTOR | P42345 | human | SRRM2 | Q9UQ35 | S1326 | NsPLRENsFGsPLEF | |
| MTOR | P42345 | human | UNK | Q9C0B0 | S255 | RKHKYRSsPCPNVKH | |
| MTOR | P42345 | human | PATL1 | Q86TB9 | S179 | ALPRRstsPIIGsPP | |
| MTOR | P42345 | human | RRAGC | Q9HB90 | T394 | kALTHNGtPRNAI__ | |
| MTOR | P42345 | human | GRB10 | Q13322 | S428 | stPVRsVsENsLVAM | BPS |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S571 | KGEDLVGsLNGGHAN | |
| MTOR | P42345 | human | SGK1 | O00141 | S422 | AEAFLGFsYAPPTDS | Pkinase_C |
| MTOR | P42345 | human | APBA3 | O96018 | S7 | _MDFPtIsRsPsGPP | |
| MTOR | P42345 | human | YAP1 | P46937 | S436 | INQSTLPsQQNRFPD | |
| MTOR | P42345 | human | ZNRF2 | Q8NHG8 | S145 | GPRLVIGsLPAHLsP | |
| MTOR | P42345 | human | RPS6 | P62753 | S236 | AKRRRLssLRAstsK | |
| MTOR | P42345 | human | RPTOR | Q8N122 | S863 | LtQsAPAsPtNkGVH | |
| MTOR | P42345 | human | OXSR1 | O95747 | S339 | EDGGWEWsDDEFDEE | |
| MTOR | P42345 | human | ISCU | Q9H1K1 | S14 | FRLRRAAsALLLRsP | |
| MTOR | P42345 | human | PATL1 | Q86TB9 | S184 | stsPIIGsPPVRAVP | |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S550 | KItsLSssLDTsLDF | |
| MTOR | P42345 | human | BAG3 | O95817 | S289 | RsstPLHsPsPIRVH | |
| MTOR | P42345 | human | DEPTOR | Q8TB45 | S286 | ssMssCGssGyFsss | |
| MTOR | P42345 | human | IRS1 | P35568 | S639 | yMPMsPKsVSAPQQI | |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | T37 | PPGDysttPGGtLFs | eIF_4EBP |
| MTOR | P42345 | human | HSF1 | Q00613 | S303 | RVkEEPPsPPQsPRV | Vert_HS_TF |
| MTOR | P42345 | human | RPS6KB1 | P23443 | S434 | sFEPKIRsPRRFIGs | |
| MTOR | P42345 | human | MYC | P01106 | S77 | LLPtPPLsPsRRsGL | Myc_N |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | T41 | ysttPGGtLFsttPG | eIF_4EBP |
| MTOR | P42345 | human | DUSP10 | Q9Y6W6 | S230 | DsFKRIFsKEIIVyD | Rhodanese |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S522 | QyktPPPsyNSALAQ | |
| MTOR | P42345 | human | AKT1S1 | Q96B36 | S221 | DLDRIAAsMRALVLR | PRAS |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S508 | KGHRKRAssENERLQ | |
| MTOR | P42345 | human | UNK | Q9C0B0 | S359 | DSVPVsPssPHAPDL | |
| MTOR | P42345 | human | DEPTOR | Q8TB45 | S265 | stsFMsVsPsKEIKI | |
| MTOR | P42345 | human | PIP4K2C | Q8TBX8 | S328 | LVGsYGTsPEGIGGY | PIP5K |
| MTOR | P42345 | human | AMBRA1 | Q9C0C7 | S52 | KRVELPDsPRSTFLL | |
| MTOR | P42345 | human | ANKRD17 | O75179 | S2045 | yPVssPssPsPPAQP | |
| MTOR | P42345 | human | HSF1 | Q00613 | S326 | ssVDtLLsPTALIDs | Vert_HS_TF |
| MTOR | P42345 | human | UBR4 | Q5T4S7 | S2932 | GHPAGPGsVsSStGA | |
| MTOR | P42345 | human | RUBCNL | Q9H714 | S157 | sPGILATsPYPETDS | |
| MTOR | P42345 | human | SCYL1 | Q96KG9 | S754 | sTQPRPDsWGEDNWE | |
| MTOR | P42345 | human | RPS6KB1 | P23443-2 | T389 | NQVFLGFtYVAPSVL | Pkinase_C |
| MTOR | P42345 | human | EIF4EBP1 | Q13541 | T46 | GGtLFsttPGGtRII | eIF_4EBP |
| MTOR | P42345 | human | ZNF768 | Q9H5H4 | S139 | sPGyEPRsPGyEsES | RNA_pol_Rpb1_R |
| MTOR | P42345 | human | SENP3 | Q9H4L4 | T142 | MLLYsKstsLtFHWK | |
| MTOR | P42345 | human | LARP6 | Q9BRS8 | S348 | DPESNPTsPMAGRRH | |
| MTOR | P42345 | human | RPS6 | P62753 | S235 | IAKRRRLssLRAsts | |
| MTOR | P42345 | human | NRBF2 | Q96F24 | S113 | AEDAEGQsPLsQkYs | NRBF2 |
| MTOR | P42345 | human | MAF1 | Q9H063 | S75 | sPsRLsksQGGEEEG | Maf1 |
| MTOR | P42345 | human | MAF1 | Q9H063 | S68 | PPQtsGLsPsRLsks | Maf1 |
| MTOR | P42345 | human | APBA3 | O96018 | T5 | ___MDFPtIsRsPsG | |
| MTOR | P42345 | human | UVRAG | Q9P2Y5 | S493 | GGADVGFsGGIPsPD | |
| MTOR | P42345 | human | PASK | Q96RG2 | S949 | RTRLFLAsLPGstHs | |
| MTOR | P42345 | human | PIP4K2C | Q8TBX8 | S324 | GPPALVGsYGTsPEG | PIP5K |
| MTOR | P42345 | human | GRB10 | Q13322 | T155 | PGsPPVLtPGsLPPS | |
| MTOR | P42345 | human | DEPTOR | Q8TB45 | S299 | ssPtLsssPPVLCNP | |
| MTOR | P42345 | human | HOXB13 | Q92826 | T8 | MEPGNYAtLDGAKDI | |
| MTOR | P42345 | human | MTOR | P42345 | T2474 | AHKKtGttVPEsIHs | |
| MTOR | P42345 | human | WNK1 | Q9H4A3 | S2032 | DGsGsPHsPHQLssK | |
| MTOR | P42345 | human | LARP1 | Q6PKG0 | S766 | EPStIARsLPttVPE | |
| MTOR | P42345 | human | PRKCE | Q02156 | T566 | LNGVTTTtFCGTPDy | Pkinase |
 Biological Network Integration of This Kinase and Substrates Biological Network Integration of This Kinase and Substrates (GeneMANIA website) |
 Enriched GO biological processes of the phosphorylation target genes of the kinase Enriched GO biological processes of the phosphorylation target genes of the kinase |
| Kinase | GOID | GO term | P.adjust |
| MTOR | ID | Description | 0.00e+00 |
| MTOR | GO:0010506 | regulation of autophagy | 2.32e-10 |
| MTOR | GO:0031929 | TOR signaling | 2.02e-08 |
| MTOR | GO:0031331 | positive regulation of cellular catabolic process | 8.18e-08 |
| MTOR | GO:0018105 | peptidyl-serine phosphorylation | 3.10e-07 |
| MTOR | GO:0018209 | peptidyl-serine modification | 4.05e-07 |
| MTOR | GO:0038202 | TORC1 signaling | 8.92e-07 |
| MTOR | GO:0010508 | positive regulation of autophagy | 1.02e-06 |
| MTOR | GO:0045931 | positive regulation of mitotic cell cycle | 4.85e-06 |
| MTOR | GO:0031667 | response to nutrient levels | 4.85e-06 |
| MTOR | GO:0010507 | negative regulation of autophagy | 7.39e-06 |
| MTOR | GO:1901654 | response to ketone | 1.12e-05 |
| MTOR | GO:0006359 | regulation of transcription by RNA polymerase III | 1.26e-05 |
| MTOR | GO:0016241 | regulation of macroautophagy | 1.72e-05 |
| MTOR | GO:0062197 | cellular response to chemical stress | 2.97e-05 |
| MTOR | GO:0031669 | cellular response to nutrient levels | 3.52e-05 |
| MTOR | GO:0016236 | macroautophagy | 3.52e-05 |
| MTOR | GO:0016239 | positive regulation of macroautophagy | 3.52e-05 |
| MTOR | GO:0000082 | G1/S transition of mitotic cell cycle | 3.52e-05 |
| MTOR | GO:0045787 | positive regulation of cell cycle | 3.54e-05 |
| MTOR | GO:0046627 | negative regulation of insulin receptor signaling pathway | 3.85e-05 |
| MTOR | GO:0006109 | regulation of carbohydrate metabolic process | 3.85e-05 |
| MTOR | GO:1900077 | negative regulation of cellular response to insulin stimulus | 3.98e-05 |
| MTOR | GO:0071496 | cellular response to external stimulus | 4.05e-05 |
| MTOR | GO:0045945 | positive regulation of transcription by RNA polymerase III | 4.13e-05 |
| MTOR | GO:0043467 | regulation of generation of precursor metabolites and energy | 5.72e-05 |
| MTOR | GO:0044843 | cell cycle G1/S phase transition | 5.72e-05 |
| MTOR | GO:0031668 | cellular response to extracellular stimulus | 5.78e-05 |
| MTOR | GO:0032869 | cellular response to insulin stimulus | 5.78e-05 |
| MTOR | GO:0031330 | negative regulation of cellular catabolic process | 6.00e-05 |
| MTOR | GO:0042594 | response to starvation | 8.27e-05 |
| MTOR | GO:1990928 | response to amino acid starvation | 1.01e-04 |
| MTOR | GO:0036294 | cellular response to decreased oxygen levels | 1.05e-04 |
| MTOR | GO:0071375 | cellular response to peptide hormone stimulus | 1.05e-04 |
| MTOR | GO:0097191 | extrinsic apoptotic signaling pathway | 1.05e-04 |
| MTOR | GO:1901655 | cellular response to ketone | 1.18e-04 |
| MTOR | GO:0006383 | transcription by RNA polymerase III | 1.61e-04 |
| MTOR | GO:0071453 | cellular response to oxygen levels | 1.79e-04 |
| MTOR | GO:0062012 | regulation of small molecule metabolic process | 1.86e-04 |
| MTOR | GO:0019216 | regulation of lipid metabolic process | 2.04e-04 |
| MTOR | GO:0008361 | regulation of cell size | 2.62e-04 |
| MTOR | GO:0046626 | regulation of insulin receptor signaling pathway | 2.62e-04 |
| MTOR | GO:0032868 | response to insulin | 2.81e-04 |
| MTOR | GO:1900076 | regulation of cellular response to insulin stimulus | 2.87e-04 |
| MTOR | GO:0008286 | insulin receptor signaling pathway | 2.90e-04 |
| MTOR | GO:0043200 | response to amino acid | 2.97e-04 |
| MTOR | GO:0046777 | protein autophosphorylation | 3.48e-04 |
| MTOR | GO:1901653 | cellular response to peptide | 3.49e-04 |
| MTOR | GO:0010827 | regulation of glucose transmembrane transport | 4.09e-04 |
| MTOR | GO:0098781 | ncRNA transcription | 4.23e-04 |
Top |
Related Drugs to MTOR_MTOR |
 Drugs used for this fusion-positive patient. Drugs used for this fusion-positive patient. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Drug | Source | PMID |
 Distribution of the number of studies mentioning MTOR-MTOR and kinase inhibitors the PubMed Abstract (04-01-2024) Distribution of the number of studies mentioning MTOR-MTOR and kinase inhibitors the PubMed Abstract (04-01-2024) |
| Fusion gene - drug pair 1 | Fusion gene - drug pair 2 | PMID | Publication date | DOI | Study title |
Top |
Related Diseases to MTOR_MTOR |
 Diseases that have this fusion gene. Diseases that have this fusion gene. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Disease | Source | PMID |
 Related diseases from the literature mentioned this fusion gene and drug. Related diseases from the literature mentioned this fusion gene and drug. (PubMed, 04-01-2024) |
| MeSH ID | MeSH term |
 Diseases associated with fusion partners. Diseases associated with fusion partners. (DisGeNet 4.0) |
| Partner | Gene | Disease ID | Disease name | # pubmeds | Source |
Top |
Clinical Trials of the Found Drugs/Small Molecules |
 Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) |
 Clinical Trials from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024) |
| Fusion Gene | Kinase Inhibitor | NCT ID | Study Status | Phases | Disease | # Enrolment | Date |

