| UTHEALTH HOME ABOUT SBMI A-Z WEBMAIL INSIDE THE UNIVERSITY |

|
|||||||
|
Kinase Fusion Gene:ARID1A_RPS6KA1 |
Kinase Fusion Protein Summary |
 Kinase Fusion gene summary Kinase Fusion gene summary |
| Kinase Fusion partner gene information | Kinase Fusion gene name: ARID1A_RPS6KA1 | KinaseFusionDB ID: KFG431 | FusionGDB2.0 ID: KFG431 | Hgene | Tgene | Gene symbol | ARID1A | RPS6KA1 | Gene ID | 8289 | 6195 | |
| Gene name | AT-rich interaction domain 1A | ribosomal protein S6 kinase A1 | ||||||||||
| Synonyms | B120|BAF250|BAF250a|BM029|C1orf4|CSS2|ELD|MRD14|OSA1|P270|SMARCF1|hELD|hOSA1 | HU-1|MAPKAPK1|MAPKAPK1A|RSK|RSK1|p90Rsk | ||||||||||
| Cytomap | 1p36.11 | 1p36.11 | ||||||||||
| Type of gene | protein-coding | protein-coding | ||||||||||
| Description | AT-rich interactive domain-containing protein 1AARID domain-containing protein 1AAT rich interactive domain 1A (SWI-like)BRG1-associated factor 250aOSA1 nuclear proteinSWI-like proteinSWI/SNF complex protein p270SWI/SNF-related, matrix-associated, | ribosomal protein S6 kinase alpha-190 kDa ribosomal protein S6 kinase 1MAP kinase-activated protein kinase 1aMAPK-activated protein kinase 1aMAPKAP kinase 1aMAPKAPK-1aRSK-1S6K-alpha 1dJ590P13.1 (ribosomal protein S6 kinase, 90kD, polypeptide 1)p9 | ||||||||||
| Modification date | 20240416 | 20240403 | ||||||||||
| UniProtAcc | O14497 | Q15418 | ||||||||||
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000324856, ENST00000374152, ENST00000457599, ENST00000540690, | ENST00000526792, ENST00000374162, ENST00000488985, ENST00000530003, ENST00000531382, ENST00000374166, ENST00000374168, | |||||||||
| Context (manual curation of fusion genes in KinaseFusionDB) | PubMed: ARID1A [Title/Abstract] AND RPS6KA1 [Title/Abstract] AND fusion [Title/Abstract] | |||||||||||
| Most frequent breakpoint (based on all fusion genes of FusionGDB 2.0) | ARID1A(27102198)-RPS6KA1(26897940), # samples:3 | |||||||||||
 Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez |
| Partner | Gene | GO ID | GO term | PubMed ID |
| Hgene | ARID1A | GO:0006337 | nucleosome disassembly | 8895581 |
| Hgene | ARID1A | GO:0006338 | chromatin remodeling | 11726552 |
| Hgene | ARID1A | GO:0030520 | intracellular estrogen receptor signaling pathway | 12200431 |
| Hgene | ARID1A | GO:0030521 | androgen receptor signaling pathway | 12200431 |
| Hgene | ARID1A | GO:0042921 | glucocorticoid receptor signaling pathway | 12200431 |
| Hgene | ARID1A | GO:0045893 | positive regulation of DNA-templated transcription | 12200431 |
| Tgene | RPS6KA1 | GO:0043154 | negative regulation of cysteine-type endopeptidase activity involved in apoptotic process | 18402937 |
 Kinase Fusion gene breakpoints across ARID1A (5'-gene) Kinase Fusion gene breakpoints across ARID1A (5'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 |
 Kinase Fusion gene breakpoints across RPS6KA1 (3'-gene) Kinase Fusion gene breakpoints across RPS6KA1 (3'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 |
Top |
Kinase Fusion Gene Sample Information |
 Kinase Fusion gene information. Kinase Fusion gene information. |
 Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)* All genome coordinats were lifted-over on hg19. * Click on the break point to see the gene structure around the break point region using the UCSC Genome Browser. |
| Source | Sample | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp |
| ChimerDB4 | TCGA-AB-2981-03B | ARID1A | chr1 | 27105774 | RPS6KA1 | chr1 | 26873403 |
| ChimerDB4 | TCGA-HU-A4GH-01A | ARID1A | chr1 | 27102198 | RPS6KA1 | chr1 | 26897940 |
| ChimerDB4 | TCGA-HU-A4GH | ARID1A | chr1 | 27102198 | RPS6KA1 | chr1 | 26897939 |
| CCLE | PSS008 | ARID1A | chr1 | 27024031 | RPS6KA1 | chr1 | 26881642 |
| CCLE | MKN-45 | ARID1A | chr1 | 27059283 | RPS6KA1 | chr1 | 26897940 |
Top |
Kinase Fusion ORF Analysis |
 Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. |
| Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | Seq length (transcript) | Seq length (amino acids) |
| ENST00000324856 | ENST00000374168 | ARID1A | chr1 | 27024031 | RPS6KA1 | chr1 | 26881642 | 3784 | 862 |
| ENST00000374152 | ENST00000374168 | ARID1A | chr1 | 27105774 | RPS6KA1 | chr1 | 26873403 | 8609 | 1815 |
Top |
Kinase Fusion Amino Acid Sequences |
 For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. |
| >Henst_Tenst_Hgene_Hchr_Hbp_Tgene_Tchr_Tbp_length(fusion AA)_AAseq >ENST00000324856_ENST00000374168_ARID1A_chr1_27024031_RPS6KA1_chr1_26881642_length(amino acids)=862 MAAQVAPAAASSLGNPPPPPPSELKKAEQQQREEAGGEAAAAAAAERGEMKAAAGQESEGPAVGPPQPLGKELQDGAESNGGGGGGGAGS GGGPGAEPDLKNSNGNAGPRPALNNNLTEPPGGGGGGSSDGVGAPPHSAAAALPPPAYGFGQPYGRSPSAVAAAAAAVFHQQHGGQQSPG LAALQSGGGGGLEPYAGPQQNSHDHGFPNHQYNSYYPNRSAYPPPAPAYALSSPRGGTPGSGAAAAAGSKPPPSSSASASSSSSSFAQQR FGAMGGGGPSAAGGGTPQPTATPTLNQLLTSPSSARGYQGYPGGDYSGGPQDGGAGKGPADMASQCWGAAAAAAAAAAASGGAQQRSHHA PMSPGSSGGGGQPLARTPQFEMLTGSLPFQGKDRKETMTLILKAKLGMPQFLSTEAQSLLRALFKRNPANRLGSGPDGAEEIKRHVFYST IDWNKLYRREIKPPFKPAVAQPDDTFYFDTEFTSRTPKDSPGIPPSAGAHQLFRGFSFVATGLMEDDGKPRAPQAPLHSVVQQLHGKNLV FSDGYVVKETIGVGSYSECKRCVHKATNMEYAVKVIDKSKRDPSEEIEILLRYGQHPNIITLKDVYDDGKHVYLVTELMRGGELLDKILR QKFFSEREASFVLHTIGKTVEYLHSQGVVHRDLKPSNILYVDESGNPECLRICDFGFAKQLRAENGLLMTPCYTANFVAPEVLKRQGYDE GCDIWSLGILLYTMLAGYTPFANGPSDTPEEILTRIGSGKFTLSGGNWNTVSETAKDLVSKMLHVDPHQRLTAKQVLQHPWVTQKDKLPQ -------------------------------------------------------------- >ENST00000374152_ENST00000374168_ARID1A_chr1_27105774_RPS6KA1_chr1_26873403_length(amino acids)=1815 MDQMGKMRPQPYGGTNPYSQQQGPPSGPQQGHGYPGQPYGSQTPQRYPMTMQGRAQSAMGGLSYTQQIPPYGQQGPSGYGQQGQTPYYNQ QSPHPQQQQPPYSQQPPSQTPHAQPSYQQQPQSQPPQLQSSQPPYSQQPSQPPHQQSPAPYPSQQSTTQQHPQSQPPYSQPQAQSPYQQQ QPQQPAPSTLSQQAAYPQPQSQQSQQTAYSQQRFPPPQELSQDSFGSQASSAPSMTSSKGGQEDMNLSLQSRPSSLPDLSGSIDDLPMGT EGALSPGVSTSGISSSQGEQSNPAQSPFSPHTSPHLPGIRGPSPSPVGSPASVAQSRSGPLSPAAVPGNQMPPRPPSGQSDSIMHPSMNQ SSIAQDRGYMQRNPQMPQYSSPQPGSALSPRQPSGGQIHTGMGSYQQNSMGSYGPQGGQYGPQGGYPRQPNYNALPNANYPSAGMAGGIN PMGAGGQMHGQPGIPPYGTLPPGRMSHASMGNRPYGPNMANMPPQVGSGMCPPPGGMNRKTQETAVAMHVAANSIQNRPPGYPNMNQGGM MGTGPPYGQGINSMAGMINPQGPPYSMGGTMANNSAGMAASPEMMGLGDVKLTPATKMNNKADGTPKTESKSKKSSSSTTTNEKITKLYE LGGEPERKMWVDRYLAFTEEKAMGMTNLPAVGRKPLDLYRLYVSVKEIGGLTQVNKNKKWRELATNLNVGTSSSAASSLKKQYIQCLYAF ECKIERGEDPPPDIFAAADSKKSQPKIQPPSPAGSGSMQGPQTPQSTSSSMAEGGDLKPPTPASTPHSQIPPLPGMSRSNSVGIQDAFND GSDSTFQKRNSMTPNPGYQPSMNTSDMMGRMSYEPNKDPYGSMRKAPGSDPFMSSGQGPNGGMGDPYSRAAGPGLGNVAMGPRQHYPYGG PYDRVRTEPGIGPEGNMSTGAPQPNLMPSNPDSGMYSPSRYPPQQQQQQQQRHDSYGNQFSTQGTPSGSPFPSQQTTMYQQQQQNYKRPM DGTYGPPAKRHEGEMYSVPYSTGQGQPQQQQLPPAQPQPASQQQAAQPSPQQDVYNQYGNAYPATATAATERRPAGGPQNQFPFQFGRDR VSAPPGTNAQQNMPPQMMGGPIQASAEVAQQGTMWQGRNDMTYNYANRQSTGSAPQGPAYHGVNRTDEMLHTDQRANHEGSWPSHGTRQP PYGPSAPVPPMTRPPPSNYQPPPSMQNHIPQVSSPAPLPRPMENRTSPSKSPFLHSGMKMQKAGPPVPASHIAPAPVQPPMIRRDITFPP GSVEATQPVLKQRRRLTMKDIGTPEAWRVMMSLKSGLLAESTWALDTINILLYDDNSIMTFNLSQPASENSEEKLISKFDKLPVKIVQKN DPFVVDCSDKLGRVQEFDSGLLHWRIGGGDTTEHIQTHFESKTELLPSRPHAPCPPAPRKHVTTAEGTPGTTDQEGPPPDGPPEKRITAT MDDMLSTRSSTLTEDGAKSSEAIKESSKFPFGISPAQSHRNIKILEDEPHSKDETPLCTLLDWQDSLAKRCVCVSNTIRSLSFVPGNDFE MSKHPGLLLILGKLILLHHKHPERKQAPLTYEKEEEQDQGVSCNKVEWWWDCLEMLRENTLVTLANISGQLDLSPYPESICLPVLDGLLH WAVCPSAEAQDPFSTLGPNAVLSPQRLVLETLSKLSIQDNNVDLILATPPFSRLEKLYSTMVRFLSDRKNPVCREMAVVLLANLAQGDSL AARAIAVQKGSIGNLLGFLEDSLAATQFQQSQASLLHMQNPPFEPTSVDMMRRAARALLALAKVDENHSEFTLYESRLLDISVSPLMNSL -------------------------------------------------------------- |
Multiple Sequence Alignment of All Fusion Protein Isoforms |
Top |
Kinase Fusion Protein Functional Features |
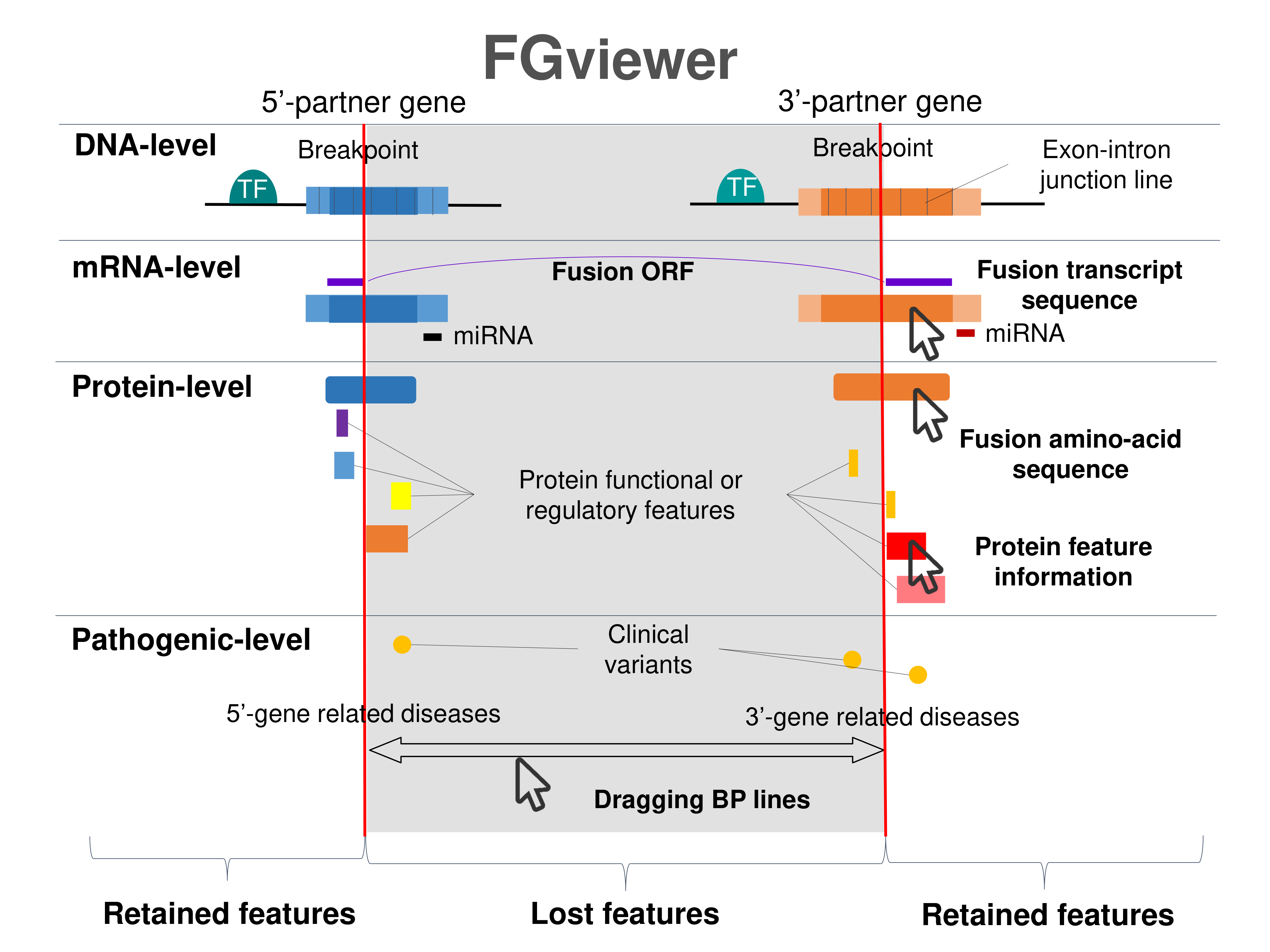
 Four levels of functional features of fusion genes Four levels of functional features of fusion genesGo to FGviewer search page for the most frequent breakpoint (https://ccsmweb.uth.edu/FGviewer/chr1:27102198/chr1:26897940) - FGviewer provides the online visualization of the retention search of the protein functional features across DNA, RNA, protein, and pathological levels. - How to search 1. Put your fusion gene symbol. 2. Press the tab key until there will be shown the breakpoint information filled. 4. Go down and press 'Search' tab twice. 4. Go down to have the hyperlink of the search result. 5. Click the hyperlink. 6. See the FGviewer result for your fusion gene. |
 |
 Main function of each fusion partner protein. (from UniProt) Main function of each fusion partner protein. (from UniProt) |
| Hgene | Tgene |
| ARID1A | RPS6KA1 |
| FUNCTION: Involved in transcriptional activation and repression of select genes by chromatin remodeling (alteration of DNA-nucleosome topology). Component of SWI/SNF chromatin remodeling complexes that carry out key enzymatic activities, changing chromatin structure by altering DNA-histone contacts within a nucleosome in an ATP-dependent manner. Binds DNA non-specifically. Belongs to the neural progenitors-specific chromatin remodeling complex (npBAF complex) and the neuron-specific chromatin remodeling complex (nBAF complex). During neural development a switch from a stem/progenitor to a postmitotic chromatin remodeling mechanism occurs as neurons exit the cell cycle and become committed to their adult state. The transition from proliferating neural stem/progenitor cells to postmitotic neurons requires a switch in subunit composition of the npBAF and nBAF complexes. As neural progenitors exit mitosis and differentiate into neurons, npBAF complexes which contain ACTL6A/BAF53A and PHF10/BAF45A, are exchanged for homologous alternative ACTL6B/BAF53B and DPF1/BAF45B or DPF3/BAF45C subunits in neuron-specific complexes (nBAF). The npBAF complex is essential for the self-renewal/proliferative capacity of the multipotent neural stem cells. The nBAF complex along with CREST plays a role regulating the activity of genes essential for dendrite growth (By similarity). {ECO:0000250|UniProtKB:A2BH40, ECO:0000303|PubMed:12672490, ECO:0000303|PubMed:22952240, ECO:0000303|PubMed:26601204}. | FUNCTION: Serine/threonine-protein kinase that acts downstream of ERK (MAPK1/ERK2 and MAPK3/ERK1) signaling and mediates mitogenic and stress-induced activation of the transcription factors CREB1, ETV1/ER81 and NR4A1/NUR77, regulates translation through RPS6 and EIF4B phosphorylation, and mediates cellular proliferation, survival, and differentiation by modulating mTOR signaling and repressing pro-apoptotic function of BAD and DAPK1 (PubMed:10679322, PubMed:16223362, PubMed:15117958, PubMed:12213813, PubMed:9430688, PubMed:17360704, PubMed:26158630, PubMed:18722121, PubMed:35772404). In fibroblast, is required for EGF-stimulated phosphorylation of CREB1, which results in the subsequent transcriptional activation of several immediate-early genes (PubMed:18508509, PubMed:18813292). In response to mitogenic stimulation (EGF and PMA), phosphorylates and activates NR4A1/NUR77 and ETV1/ER81 transcription factors and the cofactor CREBBP (PubMed:12213813, PubMed:16223362). Upon insulin-derived signal, acts indirectly on the transcription regulation of several genes by phosphorylating GSK3B at 'Ser-9' and inhibiting its activity (PubMed:18508509, PubMed:18813292). Phosphorylates RPS6 in response to serum or EGF via an mTOR-independent mechanism and promotes translation initiation by facilitating assembly of the pre-initiation complex (PubMed:17360704). In response to insulin, phosphorylates EIF4B, enhancing EIF4B affinity for the EIF3 complex and stimulating cap-dependent translation (PubMed:16763566). Is involved in the mTOR nutrient-sensing pathway by directly phosphorylating TSC2 at 'Ser-1798', which potently inhibits TSC2 ability to suppress mTOR signaling, and mediates phosphorylation of RPTOR, which regulates mTORC1 activity and may promote rapamycin-sensitive signaling independently of the PI3K/AKT pathway (PubMed:15342917). Also involved in feedback regulation of mTORC1 and mTORC2 by phosphorylating DEPTOR (PubMed:22017876). Mediates cell survival by phosphorylating the pro-apoptotic proteins BAD and DAPK1 and suppressing their pro-apoptotic function (PubMed:10679322, PubMed:16213824). Promotes the survival of hepatic stellate cells by phosphorylating CEBPB in response to the hepatotoxin carbon tetrachloride (CCl4) (PubMed:11684016). Mediates induction of hepatocyte prolifration by TGFA through phosphorylation of CEBPB (PubMed:18508509, PubMed:18813292). Is involved in cell cycle regulation by phosphorylating the CDK inhibitor CDKN1B, which promotes CDKN1B association with 14-3-3 proteins and prevents its translocation to the nucleus and inhibition of G1 progression (PubMed:18508509, PubMed:18813292). Phosphorylates EPHA2 at 'Ser-897', the RPS6KA-EPHA2 signaling pathway controls cell migration (PubMed:26158630). In response to mTORC1 activation, phosphorylates EIF4B at 'Ser-406' and 'Ser-422' which stimulates bicarbonate cotransporter SLC4A7 mRNA translation, increasing SLC4A7 protein abundance and function (PubMed:35772404). {ECO:0000269|PubMed:10679322, ECO:0000269|PubMed:11684016, ECO:0000269|PubMed:12213813, ECO:0000269|PubMed:15117958, ECO:0000269|PubMed:15342917, ECO:0000269|PubMed:16213824, ECO:0000269|PubMed:16223362, ECO:0000269|PubMed:16763566, ECO:0000269|PubMed:17360704, ECO:0000269|PubMed:18722121, ECO:0000269|PubMed:22017876, ECO:0000269|PubMed:26158630, ECO:0000269|PubMed:35772404, ECO:0000269|PubMed:9430688, ECO:0000303|PubMed:18508509, ECO:0000303|PubMed:18813292}.; FUNCTION: (Microbial infection) Promotes the late transcription and translation of viral lytic genes during Kaposi's sarcoma-associated herpesvirus/HHV-8 infection, when constitutively activated. {ECO:0000269|PubMed:30842327}. |
 Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. |
 - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
 - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 7 | 21 | 322_391 | 160 | 644 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 7 | 21 | 322_391 | 261 | 745 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 8 | 22 | 322_391 | 160 | 644 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 8 | 22 | 322_391 | 252 | 736 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 322_391 | 0 | 644 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 322_391 | 0 | 745 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 322_391 | 0 | 644 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 322_391 | 0 | 736 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 62_321 | 0 | 644 | Domain | Note=Protein kinase 1;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 62_321 | 0 | 745 | Domain | Note=Protein kinase 1;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 62_321 | 0 | 644 | Domain | Note=Protein kinase 1;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 62_321 | 0 | 736 | Domain | Note=Protein kinase 1;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 7 | 21 | 418_675 | 160 | 644 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 7 | 21 | 418_675 | 261 | 745 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 8 | 22 | 418_675 | 160 | 644 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27024031 | RPS6KA1 | 26881642 | ENST00000324856 | 8 | 22 | 418_675 | 252 | 736 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 418_675 | 0 | 644 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 21 | 418_675 | 0 | 745 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 418_675 | 0 | 644 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Tgene | ARID1A | 27105774 | RPS6KA1 | 26873403 | ENST00000374152 | 0 | 22 | 418_675 | 0 | 736 | Domain | Note=Protein kinase 2;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
Top |
Kinase Fusion Protein Structures |
 CIF files of the predicted kinase fusion proteins CIF files of the predicted kinase fusion proteins * Here we show the 3D structure of the fusion proteins using Mol*. AlphaFold produces a per-residue confidence score (pLDDT) between 0 and 100. Model confidence is shown from the pLDDT values per residue. pLDDT corresponds to the model’s prediction of its score on the local Distance Difference Test. It is a measure of local accuracy (from AlphfaFold website). To color code individual residues, we transformed individual PDB files into CIF format. |
| Kinase Fusion protein CIF link (fusion AA seq ID in KinaseFusionDB) | Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | AA seq | Len(AA seq) |
| PDB file >>>36_ARID1A_RPS6KA1 | ENST00000374152 | ENST00000374168 | ARID1A | chr1 | 27105774 | RPS6KA1 | chr1 | 26873403 | MDQMGKMRPQPYGGTNPYSQQQGPPSGPQQGHGYPGQPYGSQTPQRYPMTMQGRAQSAMGGLSYTQQIPPYGQQGPSGYGQQGQTPYYNQ QSPHPQQQQPPYSQQPPSQTPHAQPSYQQQPQSQPPQLQSSQPPYSQQPSQPPHQQSPAPYPSQQSTTQQHPQSQPPYSQPQAQSPYQQQ QPQQPAPSTLSQQAAYPQPQSQQSQQTAYSQQRFPPPQELSQDSFGSQASSAPSMTSSKGGQEDMNLSLQSRPSSLPDLSGSIDDLPMGT EGALSPGVSTSGISSSQGEQSNPAQSPFSPHTSPHLPGIRGPSPSPVGSPASVAQSRSGPLSPAAVPGNQMPPRPPSGQSDSIMHPSMNQ SSIAQDRGYMQRNPQMPQYSSPQPGSALSPRQPSGGQIHTGMGSYQQNSMGSYGPQGGQYGPQGGYPRQPNYNALPNANYPSAGMAGGIN PMGAGGQMHGQPGIPPYGTLPPGRMSHASMGNRPYGPNMANMPPQVGSGMCPPPGGMNRKTQETAVAMHVAANSIQNRPPGYPNMNQGGM MGTGPPYGQGINSMAGMINPQGPPYSMGGTMANNSAGMAASPEMMGLGDVKLTPATKMNNKADGTPKTESKSKKSSSSTTTNEKITKLYE LGGEPERKMWVDRYLAFTEEKAMGMTNLPAVGRKPLDLYRLYVSVKEIGGLTQVNKNKKWRELATNLNVGTSSSAASSLKKQYIQCLYAF ECKIERGEDPPPDIFAAADSKKSQPKIQPPSPAGSGSMQGPQTPQSTSSSMAEGGDLKPPTPASTPHSQIPPLPGMSRSNSVGIQDAFND GSDSTFQKRNSMTPNPGYQPSMNTSDMMGRMSYEPNKDPYGSMRKAPGSDPFMSSGQGPNGGMGDPYSRAAGPGLGNVAMGPRQHYPYGG PYDRVRTEPGIGPEGNMSTGAPQPNLMPSNPDSGMYSPSRYPPQQQQQQQQRHDSYGNQFSTQGTPSGSPFPSQQTTMYQQQQQNYKRPM DGTYGPPAKRHEGEMYSVPYSTGQGQPQQQQLPPAQPQPASQQQAAQPSPQQDVYNQYGNAYPATATAATERRPAGGPQNQFPFQFGRDR VSAPPGTNAQQNMPPQMMGGPIQASAEVAQQGTMWQGRNDMTYNYANRQSTGSAPQGPAYHGVNRTDEMLHTDQRANHEGSWPSHGTRQP PYGPSAPVPPMTRPPPSNYQPPPSMQNHIPQVSSPAPLPRPMENRTSPSKSPFLHSGMKMQKAGPPVPASHIAPAPVQPPMIRRDITFPP GSVEATQPVLKQRRRLTMKDIGTPEAWRVMMSLKSGLLAESTWALDTINILLYDDNSIMTFNLSQPASENSEEKLISKFDKLPVKIVQKN DPFVVDCSDKLGRVQEFDSGLLHWRIGGGDTTEHIQTHFESKTELLPSRPHAPCPPAPRKHVTTAEGTPGTTDQEGPPPDGPPEKRITAT MDDMLSTRSSTLTEDGAKSSEAIKESSKFPFGISPAQSHRNIKILEDEPHSKDETPLCTLLDWQDSLAKRCVCVSNTIRSLSFVPGNDFE MSKHPGLLLILGKLILLHHKHPERKQAPLTYEKEEEQDQGVSCNKVEWWWDCLEMLRENTLVTLANISGQLDLSPYPESICLPVLDGLLH WAVCPSAEAQDPFSTLGPNAVLSPQRLVLETLSKLSIQDNNVDLILATPPFSRLEKLYSTMVRFLSDRKNPVCREMAVVLLANLAQGDSL AARAIAVQKGSIGNLLGFLEDSLAATQFQQSQASLLHMQNPPFEPTSVDMMRRAARALLALAKVDENHSEFTLYESRLLDISVSPLMNSL | 1815 |
| 3D view using mol* of 36_ARID1A_RPS6KA1 | ||||||||||
| PDB file >>>TKFP_55_ARID1A_RPS6KA1 | ENST00000324856 | ENST00000374168 | ARID1A | chr1 | 27024031 | RPS6KA1 | chr1 | 26881642 | MAAQVAPAAASSLGNPPPPPPSELKKAEQQQREEAGGEAAAAAAAERGEMKAAAGQESEGPAVGPPQPLGKELQDGAESNGGGGGGGAGS GGGPGAEPDLKNSNGNAGPRPALNNNLTEPPGGGGGGSSDGVGAPPHSAAAALPPPAYGFGQPYGRSPSAVAAAAAAVFHQQHGGQQSPG LAALQSGGGGGLEPYAGPQQNSHDHGFPNHQYNSYYPNRSAYPPPAPAYALSSPRGGTPGSGAAAAAGSKPPPSSSASASSSSSSFAQQR FGAMGGGGPSAAGGGTPQPTATPTLNQLLTSPSSARGYQGYPGGDYSGGPQDGGAGKGPADMASQCWGAAAAAAAAAAASGGAQQRSHHA PMSPGSSGGGGQPLARTPQFEMLTGSLPFQGKDRKETMTLILKAKLGMPQFLSTEAQSLLRALFKRNPANRLGSGPDGAEEIKRHVFYST IDWNKLYRREIKPPFKPAVAQPDDTFYFDTEFTSRTPKDSPGIPPSAGAHQLFRGFSFVATGLMEDDGKPRAPQAPLHSVVQQLHGKNLV FSDGYVVKETIGVGSYSECKRCVHKATNMEYAVKVIDKSKRDPSEEIEILLRYGQHPNIITLKDVYDDGKHVYLVTELMRGGELLDKILR QKFFSEREASFVLHTIGKTVEYLHSQGVVHRDLKPSNILYVDESGNPECLRICDFGFAKQLRAENGLLMTPCYTANFVAPEVLKRQGYDE GCDIWSLGILLYTMLAGYTPFANGPSDTPEEILTRIGSGKFTLSGGNWNTVSETAKDLVSKMLHVDPHQRLTAKQVLQHPWVTQKDKLPQ | 862_ARID1A_RPS6KA1 |
| PDB file >>>TKFP_56_ARID1A_RPS6KA1 | ENST00000374152 | ENST00000374168 | ARID1A | chr1 | 27105774 | RPS6KA1 | chr1 | 26873403 | MDQMGKMRPQPYGGTNPYSQQQGPPSGPQQGHGYPGQPYGSQTPQRYPMTMQGRAQSAMGGLSYTQQIPPYGQQGPSGYGQQGQTPYYNQ QSPHPQQQQPPYSQQPPSQTPHAQPSYQQQPQSQPPQLQSSQPPYSQQPSQPPHQQSPAPYPSQQSTTQQHPQSQPPYSQPQAQSPYQQQ QPQQPAPSTLSQQAAYPQPQSQQSQQTAYSQQRFPPPQELSQDSFGSQASSAPSMTSSKGGQEDMNLSLQSRPSSLPDLSGSIDDLPMGT EGALSPGVSTSGISSSQGEQSNPAQSPFSPHTSPHLPGIRGPSPSPVGSPASVAQSRSGPLSPAAVPGNQMPPRPPSGQSDSIMHPSMNQ SSIAQDRGYMQRNPQMPQYSSPQPGSALSPRQPSGGQIHTGMGSYQQNSMGSYGPQGGQYGPQGGYPRQPNYNALPNANYPSAGMAGGIN PMGAGGQMHGQPGIPPYGTLPPGRMSHASMGNRPYGPNMANMPPQVGSGMCPPPGGMNRKTQETAVAMHVAANSIQNRPPGYPNMNQGGM MGTGPPYGQGINSMAGMINPQGPPYSMGGTMANNSAGMAASPEMMGLGDVKLTPATKMNNKADGTPKTESKSKKSSSSTTTNEKITKLYE LGGEPERKMWVDRYLAFTEEKAMGMTNLPAVGRKPLDLYRLYVSVKEIGGLTQVNKNKKWRELATNLNVGTSSSAASSLKKQYIQCLYAF ECKIERGEDPPPDIFAAADSKKSQPKIQPPSPAGSGSMQGPQTPQSTSSSMAEGGDLKPPTPASTPHSQIPPLPGMSRSNSVGIQDAFND GSDSTFQKRNSMTPNPGYQPSMNTSDMMGRMSYEPNKDPYGSMRKAPGSDPFMSSGQGPNGGMGDPYSRAAGPGLGNVAMGPRQHYPYGG PYDRVRTEPGIGPEGNMSTGAPQPNLMPSNPDSGMYSPSRYPPQQQQQQQQRHDSYGNQFSTQGTPSGSPFPSQQTTMYQQQQQNYKRPM DGTYGPPAKRHEGEMYSVPYSTGQGQPQQQQLPPAQPQPASQQQAAQPSPQQDVYNQYGNAYPATATAATERRPAGGPQNQFPFQFGRDR VSAPPGTNAQQNMPPQMMGGPIQASAEVAQQGTMWQGRNDMTYNYANRQSTGSAPQGPAYHGVNRTDEMLHTDQRANHEGSWPSHGTRQP PYGPSAPVPPMTRPPPSNYQPPPSMQNHIPQVSSPAPLPRPMENRTSPSKSPFLHSGMKMQKAGPPVPASHIAPAPVQPPMIRRDITFPP GSVEATQPVLKQRRRLTMKDIGTPEAWRVMMSLKSGLLAESTWALDTINILLYDDNSIMTFNLSQPASENSEEKLISKFDKLPVKIVQKN DPFVVDCSDKLGRVQEFDSGLLHWRIGGGDTTEHIQTHFESKTELLPSRPHAPCPPAPRKHVTTAEGTPGTTDQEGPPPDGPPEKRITAT MDDMLSTRSSTLTEDGAKSSEAIKESSKFPFGISPAQSHRNIKILEDEPHSKDETPLCTLLDWQDSLAKRCVCVSNTIRSLSFVPGNDFE MSKHPGLLLILGKLILLHHKHPERKQAPLTYEKEEEQDQGVSCNKVEWWWDCLEMLRENTLVTLANISGQLDLSPYPESICLPVLDGLLH WAVCPSAEAQDPFSTLGPNAVLSPQRLVLETLSKLSIQDNNVDLILATPPFSRLEKLYSTMVRFLSDRKNPVCREMAVVLLANLAQGDSL AARAIAVQKGSIGNLLGFLEDSLAATQFQQSQASLLHMQNPPFEPTSVDMMRRAARALLALAKVDENHSEFTLYESRLLDISVSPLMNSL | 1815_ARID1A_RPS6KA1 |
| 3D view using mol* of TKFP_56_ARID1A_RPS6KA1 | ||||||||||
Top |
Comparison of Fusion Protein Isoforms |
 Superimpose the 3D Structures Among All Fusion Protein Isoforms Superimpose the 3D Structures Among All Fusion Protein Isoforms * Download the pdb file and open it from the molstar online viewer. |
| 3D view using mol* of viewer/superimpose_isoforms/TKFP_55_ARID1A_RPS6KA1_vs_TKFP_56_ARID1A_RPS6KA1_superimposed.pdb.html |
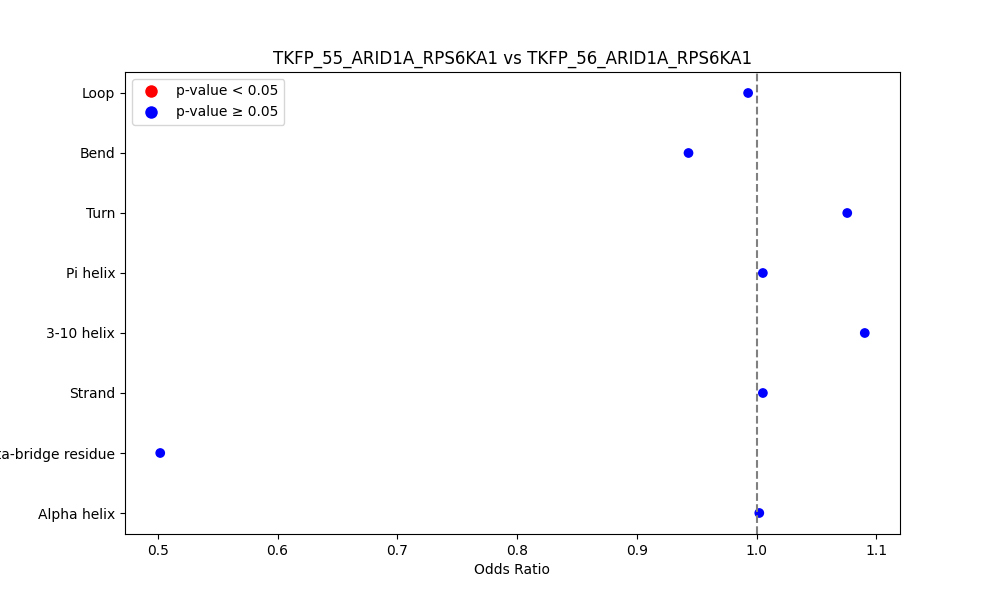
 Comparison of the Secondary Structures of Fusion Protein Isoforms Comparison of the Secondary Structures of Fusion Protein Isoforms |
| ./secondary_str/TKFP_55_ARID1A_RPS6KA1_vs_TKFP_56_ARID1A_RPS6KA1.png |
 |
Top |
Comparison of Fusion Protein Sequences/Structures with Known Sequences/Structures from PDB |
Top |
pLDDT score distribution |
 pLDDT score distribution of the predicted fusion protein structures from AlphaFold2 pLDDT score distribution of the predicted fusion protein structures from AlphaFold2* AlphaFold produces a per-residue confidence score (pLDDT) between 0 and 100. * The blue color at the bottom marks the best active site residues. |
| 36_ARID1A_RPS6KA1.png |
 |
| 36_ARID1A_RPS6KA1.png |
 |
| TKFP_56_ARID1A_RPS6KA1.png |
 |
Top |
Potential Active Site Information |
 The potential binding sites of these fusion proteins were identified using SiteMap, a module of the Schrodinger suite. The potential binding sites of these fusion proteins were identified using SiteMap, a module of the Schrodinger suite. |
| Kinase Fusion AA seq ID in KinaseFusionDB | Site score | Size | Dscore | Volume | Exposure | Enclosure | Contact | Phobic | Philic | Balance | Don/Acc | Residues |
| TKFP_56_ARID1A_RPS6KA1 | 1.096 | 120 | 1.187 | 499.408 | 0.716 | 0.69 | 0.787 | 1.371 | 0.514 | 2.666 | 1.283 | Chain A: 271,726,727,728,1293,1296,1301,1305,1385, 1386,1387,1392,1501,1503,1504,1507,1508,1510,1511, 1514,1515,1518,1537,1540,1541,1544,1547,1548,1575, 1576,1578,1579,1582,1583,1586,1589,1590,1593 |
Top |
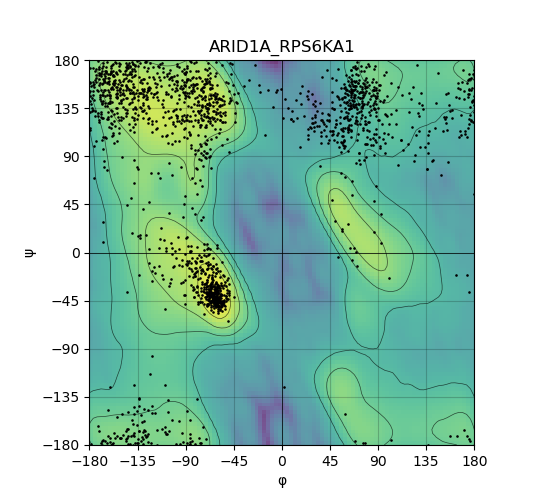
Ramachandran Plot of Kinase Fusion Protein Structure |
 Ramachandran plot of the torsional angles - phi (φ)and psi (ψ) - of the residues (amino acids) contained in this fusion protein peptide. Ramachandran plot of the torsional angles - phi (φ)and psi (ψ) - of the residues (amino acids) contained in this fusion protein peptide. |
| 36_ARID1A_RPS6KA1_ramachandran.png |
 |
| TKFP_56_ARID1A_RPS6KA1_ramachandran.png |
 |
Top |
Virtual Screening Results |
 Distribution of the average docking score across all approved kinase inhibitors. Distribution of the average docking score across all approved kinase inhibitors.Distribution of the number of occurrence across all approved kinase inhibitors. |
| 5'-kinase fusion protein case |
| 3'-kinase fusion protein case |
Top |
 Drug information from DrugBank of the top 20 interacting small molecules. Drug information from DrugBank of the top 20 interacting small molecules.* The detailed information of individual kinase inhibitors are available in the download page. |
| Fusion gene name info | Drug | Docking score | Glide g score | Glide energy |
Top |
Kinase-Substrate Information of ARID1A_RPS6KA1 |
 Phosphorylation target of the kinase Phosphorylation target of the kinase(phosphosite, 03-17-2024) |
| Kinase | Kinase UniProt Acc | Kinase species | Substrate | Substrate UniProt Acc | Substrate phosphorylated residues | Substrate phosphorylated sites (+/-7AA) | Domain |
| RPS6KA1 | Q15418 | human | TERF2IP | Q9NYB0 | S205 | GDAPVsPssQkLKRK | |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S318 | EFRsRCssVtGVQRR | PID |
| RPS6KA1 | Q15418 | human | SLC9A1 | P19634 | S703 | MsRARIGsDPLAyEP | |
| RPS6KA1 | Q15418 | human | WWC1 | Q8IX03 | S947 | CRLNRsDsDSSTLSK | |
| RPS6KA1 | Q15418 | human | NDRG2 | Q9UN36 | S350 | RsRsRtLsQssEsGt | |
| RPS6KA1 | Q15418 | human | VASP | P50552 | T278 | LARRRKAtQVGEktP | |
| RPS6KA1 | Q15418 | human | RPS6KA1 | Q15418 | K335 | KLYRREIkPPFKPAV | |
| RPS6KA1 | Q15418 | human | CDC25B | P30305 | T355 | NkRRRsVtPPEEQQE | M-inducer_phosp |
| RPS6KA1 | Q15418 | human | REPS1 | Q96D71 | S709 | PVRRRLksEDELRPE | |
| RPS6KA1 | Q15418 | human | CDC25C | P30307 | S247 | kVKKkYFsGQGkLRK | M-inducer_phosp |
| RPS6KA1 | Q15418 | human | RIOK2 | Q9BVS4 | S483 | QYRTRtLsItssGsA | |
| RPS6KA1 | Q15418 | human | RPTOR | Q8N122 | S721 | tPrLrsVssyGNIRA | |
| RPS6KA1 | Q15418 | human | EEF2K | O00418 | S366 | sPQVRtLsGSRPPLL | |
| RPS6KA1 | Q15418 | human | MXD1 | Q05195 | S145 | IERIRMDsIGSTVSS | |
| RPS6KA1 | Q15418 | human | YBX1 | P67809 | S102 | NPRKyLrsVGDGEtV | CSD |
| RPS6KA1 | Q15418 | human | RPS6KA1 | Q15418 | S380 | HQLFRGFsFVAtGLM | Pkinase_C |
| RPS6KA1 | Q15418 | human | PDCD4 | Q53EL6 | S457 | RGRKRFVsEGDGGRL | |
| RPS6KA1 | Q15418 | human | SOS1 | Q07889 | S1134 | PHGPRsAsVssISLT | |
| RPS6KA1 | Q15418 | human | LCP1 | P13796 | S5 | ___MARGsVsDEEMM | |
| RPS6KA1 | Q15418 | human | SOS1 | Q07889 | S1161 | PPRRRPEsAPAEssP | |
| RPS6KA1 | Q15418 | human | DAPK1 | P53355 | S289 | QALSRKAsAVNMEkF | |
| RPS6KA1 | Q15418 | human | METTL1 | Q9UBP6 | S27 | YYRQrAHsNPMADHT | |
| RPS6KA1 | Q15418 | human | OSTF1 | Q92882 | S202 | TDAVRtLsNAEDyLD | |
| RPS6KA1 | Q15418 | human | RPS6KA1 | Q15418 | K110 | VRDRVRTkMERDILA | |
| RPS6KA1 | Q15418 | human | CDC25A | P30304 | S295 | TKrRKsMsGASPkES | M-inducer_phosp |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S570 | AKRsLtssLENIFsr | |
| RPS6KA1 | Q15418 | human | EPHA2 | P29317 | S897 | RVsIRLPstsGsEGV | |
| RPS6KA1 | Q15418 | human | EIF4B | P23588 | S425 | sRtGsEssQtGtstt | |
| RPS6KA1 | Q15418 | human | ITGB4 | P16144 | S1364 | PSGsQRPsVSDDTGC | |
| RPS6KA1 | Q15418 | human | FOS | P01100 | S362 | AAAHRKGssSNEPSS | |
| RPS6KA1 | Q15418 | human | WWC1 | Q8IX03 | T929 | stIIRsKtFsPGPQS | |
| RPS6KA1 | Q15418 | human | NFKBIA | P25963 | S32 | LLDDRHDsGLDsMkD | |
| RPS6KA1 | Q15418 | human | APAF1 | O14727 | S268 | LLTTRDksVTDsVMG | NB-ARC |
| RPS6KA1 | Q15418 | human | CDC25B | P30305 | S353 | VQNkRRRsVtPPEEQ | M-inducer_phosp |
| RPS6KA1 | Q15418 | human | UBR5 | O95071 | S2483 | ENRKRHGssRsVVDM | |
| RPS6KA1 | Q15418 | human | MAPK7 | Q13164 | S496 | SRLRDGPsAPLEAPE | |
| RPS6KA1 | Q15418 | human | BAD | Q92934 | S75 | EIRsRHssyPAGtED | Bcl-2_BAD |
| RPS6KA1 | Q15418 | human | RPS6KA1 | Q15418 | K421 | FSDGYVVkETIGVGS | |
| RPS6KA1 | Q15418 | human | RPTOR | Q8N122 | S719 | PCtPrLrsVssyGNI | |
| RPS6KA1 | Q15418 | human | CREB1 | P16220 | S119 | EILsRRPsyRkILND | pKID |
| RPS6KA1 | Q15418 | human | ARHGAP31 | Q2M1Z3 | S1106 | KGKNRPSsLNLDPAI | |
| RPS6KA1 | Q15418 | human | GSK3B | P49841 | S9 | SGRPRttsFAEsCkP | |
| RPS6KA1 | Q15418 | human | NDRG2 | Q9UN36 | S332 | LsRsRtAsLtsAAsV | |
| RPS6KA1 | Q15418 | human | RANBP3 | Q9H6Z4 | S126 | VKRERtssLtQFPPs | |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S751 | EGRKRtsstCsNEsL | |
| RPS6KA1 | Q15418 | human | CIC | Q96RK0 | S301 | EtRERsMsEtGtAAA | |
| RPS6KA1 | Q15418 | human | DEPTOR | Q8TB45 | S287 | sMssCGssGyFsssP | |
| RPS6KA1 | Q15418 | human | UBR5 | O95071 | T637 | PYKRRRstPAPKEEE | |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S341 | QPRRRHAsAPsHVQP | PID |
| RPS6KA1 | Q15418 | human | TSC2 | P49815 | S1798 | GQRKRLIssVEDFTE | |
| RPS6KA1 | Q15418 | human | ESR1 | P03372 | S167 | GGRERLAsTNDkGSM | Oest_recep |
| RPS6KA1 | Q15418 | human | CARHSP1 | Q9Y2V2 | S52 | tRRtrtFsAtVRAsQ | |
| RPS6KA1 | Q15418 | human | NHERF1 | O14745 | T156 | ELRPRLCtMKKGPsG | PDZ |
| RPS6KA1 | Q15418 | human | RPS6 | P62753 | S236 | AKRRRLssLRAstsK | |
| RPS6KA1 | Q15418 | human | CHEK1 | O14757 | S280 | AKRPRVtsGGVsEsP | |
| RPS6KA1 | Q15418 | human | MITF | O75030-9 | S409 | KTSsRRSsMSMEETE | DUF3371 |
| RPS6KA1 | Q15418 | human | RELA | Q04206 | S276 | sMQLRRPsDRELsEP | RHD_dimer |
| RPS6KA1 | Q15418 | human | DEPTOR | Q8TB45 | S286 | ssMssCGssGyFsss | |
| RPS6KA1 | Q15418 | human | FLNA | P21333 | S2152 | tRRRRAPsVANVGsH | Filamin |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | T642 | QFRRRAHtFsHPPss | |
| RPS6KA1 | Q15418 | human | CCT2 | P78371 | S260 | GsRVRVDstAkVAEI | Cpn60_TCP1 |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | T568 | NKAKRsLtssLENIF | |
| RPS6KA1 | Q15418 | human | APAF1 | O14727 | S357 | FKRIRKSsSYDYEAL | NB-ARC |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S588 | RMRGRLGsVDsFERs | |
| RPS6KA1 | Q15418 | human | UBR5 | O95071 | S1227 | DCKLKRTsPTAYCDC | |
| RPS6KA1 | Q15418 | human | TBC1D4 | O60343 | S666 | GRAQGVRsPLLRQSS | |
| RPS6KA1 | Q15418 | human | PDCD4 | Q53EL6 | S76 | RDsGrGDsVsDsGsD | |
| RPS6KA1 | Q15418 | human | GMFB | P60983 | T27 | KFRFRKEtNNAAIIM | Cofilin_ADF |
| RPS6KA1 | Q15418 | human | CDC34 | P49427 | T162 | KGkDREYtDIIrkQV | UQ_con |
| RPS6KA1 | Q15418 | human | MAGI1 | Q96QZ7 | S741 | QPLERKDsQNSSQHs | MAGI_u5 |
| RPS6KA1 | Q15418 | human | EIF4B | P23588 | S422 | RERsRtGsEssQtGt | |
| RPS6KA1 | Q15418 | human | RPS6 | P62753 | S235 | IAKRRRLssLRAsts | |
| RPS6KA1 | Q15418 | human | SENP2 | Q9HC62 | T369 | TDDLLELtEDMEKEI | |
| RPS6KA1 | Q15418 | human | DEPTOR | Q8TB45 | S291 | CGssGyFsssPtLss | |
| RPS6KA1 | Q15418 | human | ARHGAP31 | Q2M1Z3 | S1178 | ALTGRRNsAPVsVsA | |
| RPS6KA1 | Q15418 | human | CIC | Q96RK0 | S173 | PGKRRtQsLsALPKE | |
| RPS6KA1 | Q15418 | human | CDC25A | P30304 | S293 | GSTKrRKsMsGASPk | M-inducer_phosp |
| RPS6KA1 | Q15418 | human | RPTOR | Q8N122 | S722 | PrLrsVssyGNIRAV |
 Biological Network Integration of This Kinase and Substrates Biological Network Integration of This Kinase and Substrates (GeneMANIA website) |
 Enriched GO biological processes of the phosphorylation target genes of the kinase Enriched GO biological processes of the phosphorylation target genes of the kinase |
| Kinase | GOID | GO term | P.adjust |
| RPS6KA1 | ID | Description | 0.00e+00 |
| RPS6KA1 | GO:0035994 | response to muscle stretch | 5.21e-04 |
| RPS6KA1 | GO:0032868 | response to insulin | 5.21e-04 |
| RPS6KA1 | GO:0032869 | cellular response to insulin stimulus | 6.65e-04 |
| RPS6KA1 | GO:0071375 | cellular response to peptide hormone stimulus | 6.65e-04 |
| RPS6KA1 | GO:0043434 | response to peptide hormone | 6.65e-04 |
| RPS6KA1 | GO:1901993 | regulation of meiotic cell cycle phase transition | 6.65e-04 |
| RPS6KA1 | GO:0044771 | meiotic cell cycle phase transition | 1.35e-03 |
| RPS6KA1 | GO:1901653 | cellular response to peptide | 1.51e-03 |
| RPS6KA1 | GO:0030316 | osteoclast differentiation | 1.96e-03 |
| RPS6KA1 | GO:0030011 | maintenance of cell polarity | 2.67e-03 |
| RPS6KA1 | GO:0030099 | myeloid cell differentiation | 3.03e-03 |
| RPS6KA1 | GO:0009612 | response to mechanical stimulus | 3.28e-03 |
| RPS6KA1 | GO:0046824 | positive regulation of nucleocytoplasmic transport | 3.28e-03 |
| RPS6KA1 | GO:0006913 | nucleocytoplasmic transport | 3.39e-03 |
| RPS6KA1 | GO:0051169 | nuclear transport | 3.39e-03 |
| RPS6KA1 | GO:0097191 | extrinsic apoptotic signaling pathway | 3.62e-03 |
| RPS6KA1 | GO:0044772 | mitotic cell cycle phase transition | 3.62e-03 |
| RPS6KA1 | GO:0045670 | regulation of osteoclast differentiation | 3.62e-03 |
| RPS6KA1 | GO:0014074 | response to purine-containing compound | 3.94e-03 |
| RPS6KA1 | GO:0010508 | positive regulation of autophagy | 4.28e-03 |
| RPS6KA1 | GO:0045672 | positive regulation of osteoclast differentiation | 4.28e-03 |
| RPS6KA1 | GO:0051446 | positive regulation of meiotic cell cycle | 4.28e-03 |
| RPS6KA1 | GO:0010971 | positive regulation of G2/M transition of mitotic cell cycle | 5.02e-03 |
| RPS6KA1 | GO:0036294 | cellular response to decreased oxygen levels | 5.02e-03 |
| RPS6KA1 | GO:0031929 | TOR signaling | 5.53e-03 |
| RPS6KA1 | GO:1902751 | positive regulation of cell cycle G2/M phase transition | 5.53e-03 |
| RPS6KA1 | GO:0031331 | positive regulation of cellular catabolic process | 5.53e-03 |
| RPS6KA1 | GO:0071868 | cellular response to monoamine stimulus | 5.53e-03 |
| RPS6KA1 | GO:0071870 | cellular response to catecholamine stimulus | 5.53e-03 |
| RPS6KA1 | GO:2001233 | regulation of apoptotic signaling pathway | 5.53e-03 |
| RPS6KA1 | GO:0071867 | response to monoamine | 5.76e-03 |
| RPS6KA1 | GO:0071869 | response to catecholamine | 5.76e-03 |
| RPS6KA1 | GO:0071453 | cellular response to oxygen levels | 5.76e-03 |
| RPS6KA1 | GO:1903706 | regulation of hemopoiesis | 6.52e-03 |
| RPS6KA1 | GO:1901992 | positive regulation of mitotic cell cycle phase transition | 7.20e-03 |
| RPS6KA1 | GO:0007212 | dopamine receptor signaling pathway | 7.20e-03 |
| RPS6KA1 | GO:0008361 | regulation of cell size | 7.20e-03 |
| RPS6KA1 | GO:0043620 | regulation of DNA-templated transcription in response to stress | 7.56e-03 |
| RPS6KA1 | GO:0031400 | negative regulation of protein modification process | 7.59e-03 |
| RPS6KA1 | GO:0070884 | regulation of calcineurin-NFAT signaling cascade | 7.75e-03 |
| RPS6KA1 | GO:0010389 | regulation of G2/M transition of mitotic cell cycle | 7.77e-03 |
| RPS6KA1 | GO:0034504 | protein localization to nucleus | 7.77e-03 |
| RPS6KA1 | GO:0106056 | regulation of calcineurin-mediated signaling | 7.77e-03 |
| RPS6KA1 | GO:0036293 | response to decreased oxygen levels | 8.13e-03 |
| RPS6KA1 | GO:0062197 | cellular response to chemical stress | 8.22e-03 |
| RPS6KA1 | GO:0046822 | regulation of nucleocytoplasmic transport | 8.34e-03 |
| RPS6KA1 | GO:1902105 | regulation of leukocyte differentiation | 8.34e-03 |
| RPS6KA1 | GO:0016055 | Wnt signaling pathway | 8.34e-03 |
| RPS6KA1 | GO:0198738 | cell-cell signaling by wnt | 8.38e-03 |
Top |
Related Drugs to ARID1A_RPS6KA1 |
 Drugs used for this fusion-positive patient. Drugs used for this fusion-positive patient. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Drug | Source | PMID |
 Distribution of the number of studies mentioning ARID1A-RPS6KA1 and kinase inhibitors the PubMed Abstract (04-01-2024) Distribution of the number of studies mentioning ARID1A-RPS6KA1 and kinase inhibitors the PubMed Abstract (04-01-2024) |
| Fusion gene - drug pair 1 | Fusion gene - drug pair 2 | PMID | Publication date | DOI | Study title |
Top |
Related Diseases to ARID1A_RPS6KA1 |
 Diseases that have this fusion gene. Diseases that have this fusion gene. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Disease | Source | PMID |
 Related diseases from the literature mentioned this fusion gene and drug. Related diseases from the literature mentioned this fusion gene and drug. (PubMed, 04-01-2024) |
| MeSH ID | MeSH term |
 Diseases associated with fusion partners. Diseases associated with fusion partners. (DisGeNet 4.0) |
| Partner | Gene | Disease ID | Disease name | # pubmeds | Source |
| Hgene | ARID1A | C0024623 | Malignant neoplasm of stomach | 3 | CTD_human |
| Hgene | ARID1A | C0038356 | Stomach Neoplasms | 3 | CTD_human |
| Hgene | ARID1A | C1708349 | Hereditary Diffuse Gastric Cancer | 3 | CTD_human |
| Hgene | ARID1A | C2239176 | Liver carcinoma | 3 | CTD_human |
| Hgene | ARID1A | C0033578 | Prostatic Neoplasms | 2 | CTD_human |
| Hgene | ARID1A | C0376358 | Malignant neoplasm of prostate | 2 | CTD_human |
Top |
Clinical Trials of the Found Drugs/Small Molecules |
 Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) |
 Clinical Trials from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024) |
| Fusion Gene | Kinase Inhibitor | NCT ID | Study Status | Phases | Disease | # Enrolment | Date |

