| UTHEALTH HOME ABOUT SBMI A-Z WEBMAIL INSIDE THE UNIVERSITY |

|
|||||||
|
Kinase Fusion Gene:ROCK1_LAMA3 |
Kinase Fusion Protein Summary |
 Kinase Fusion gene summary Kinase Fusion gene summary |
| Kinase Fusion partner gene information | Kinase Fusion gene name: ROCK1_LAMA3 | KinaseFusionDB ID: KFG5432 | FusionGDB2.0 ID: KFG5432 | Hgene | Tgene | Gene symbol | ROCK1 | LAMA3 | Gene ID | 6093 | 3910 | |
| Gene name | Rho associated coiled-coil containing protein kinase 1 | laminin subunit alpha 4 | ||||||||||
| Synonyms | P160ROCK|ROCK-I | CMD1JJ|LAMA3|LAMA4*-1 | ||||||||||
| Cytomap | 18q11.1 | 6q21 | ||||||||||
| Type of gene | protein-coding | protein-coding | ||||||||||
| Description | rho-associated protein kinase 1p160 ROCK-1renal carcinoma antigen NY-REN-35 | laminin subunit alpha-4laminin alpha 4 chainlaminin, alpha 4 | ||||||||||
| Modification date | 20240305 | 20240403 | ||||||||||
| UniProtAcc | Q13464 | Q16787 | ||||||||||
| Ensembl transtripts involved in fusion gene | ENST ids | ENST00000399799, | ENST00000269217, ENST00000587184, ENST00000588770, ENST00000313654, ENST00000399516, | |||||||||
| Context (manual curation of fusion genes in KinaseFusionDB) | PubMed: ROCK1 [Title/Abstract] AND LAMA3 [Title/Abstract] AND fusion [Title/Abstract] | |||||||||||
| Most frequent breakpoint (based on all fusion genes of FusionGDB 2.0) | ROCK1(18546878)-LAMA3(21393021), # samples:2 | |||||||||||
 Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology of each fusion partner gene with evidence of Inferred from Direct Assay (IDA) from Entrez |
| Partner | Gene | GO ID | GO term | PubMed ID |
| Hgene | ROCK1 | GO:0006468 | protein phosphorylation | 23093407 |
| Hgene | ROCK1 | GO:0007159 | leukocyte cell-cell adhesion | 12082081 |
| Hgene | ROCK1 | GO:0018105 | peptidyl-serine phosphorylation | 18573880 |
| Hgene | ROCK1 | GO:0022614 | membrane to membrane docking | 12082081 |
| Hgene | ROCK1 | GO:0030334 | regulation of cell migration | 23093407 |
| Hgene | ROCK1 | GO:0032091 | negative regulation of protein binding | 18573880 |
| Hgene | ROCK1 | GO:0032956 | regulation of actin cytoskeleton organization | 25911094 |
| Hgene | ROCK1 | GO:0050900 | leukocyte migration | 12082081 |
| Hgene | ROCK1 | GO:0050901 | leukocyte tethering or rolling | 12082081 |
| Hgene | ROCK1 | GO:0070507 | regulation of microtubule cytoskeleton organization | 23093407 |
 Kinase Fusion gene breakpoints across ROCK1 (5'-gene) Kinase Fusion gene breakpoints across ROCK1 (5'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 |
 Kinase Fusion gene breakpoints across LAMA3 (3'-gene) Kinase Fusion gene breakpoints across LAMA3 (3'-gene)* Click on the image to open the UCSC genome browser with custom track showing this image in a new window. |
 |
Top |
Kinase Fusion Gene Sample Information |
 Kinase Fusion gene information. Kinase Fusion gene information. |
 Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE) Kinase Fusion gene information from four resources (ChiTars 5.0, ChimerDB 4.0, COSMIC, and CCLE)* All genome coordinats were lifted-over on hg19. * Click on the break point to see the gene structure around the break point region using the UCSC Genome Browser. |
| Source | Sample | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp |
| ChimerDB4 | TCGA-CG-4301-01A | ROCK1 | chr18 | 18546878 | LAMA3 | chr18 | 21393021 |
Top |
Kinase Fusion ORF Analysis |
 Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. Kinase Fusion information from ORFfinder translation from full-length transcript sequence from KinaseFusionDB. |
| Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | Seq length (transcript) | Seq length (amino acids) |
| ENST00000399799 | ENST00000313654 | ROCK1 | chr18 | 18546878 | LAMA3 | chr18 | 21393021 | 12972 | 3884 |
Top |
Kinase Fusion Amino Acid Sequences |
 For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. For individual full-length fusion transcript sequence from KinaseFusionDB, we ran ORFfinder and chose the longest ORF among the all predicted ones. |
| >Henst_Tenst_Hgene_Hchr_Hbp_Tgene_Tchr_Tbp_length(fusion AA)_AAseq >ENST00000399799_ENST00000313654_ROCK1_chr18_18546878_LAMA3_chr18_21393021_length(amino acids)=3884 MLVLVRGHCHSSSNMSTGDSFETRFEKMDNLLRDPKSEVNSDCLLDGLDALVYDLDFPALRKNKNIDNFLSRYKDTINKIRDLRMKAEDY EVVKVIGRGAFGEVQLVRHKSTRKVYAMKLLSKFEMIKRSDSAFFWEERDIMAFANSPWVVQLFYAFQDDRYLYMVMEYMPGGDLVNLMS NYDVPEKWARFYTAEVVLALDAIHSMGFIHRDVKPDNMLLDKSGHLKLADFGTCMKMNKEGMVRCDTAVGTPDYISPEVLKSQGGDGYYG RECDWWSVGVFLYEMLVGDTPFYADSLVGTYSKIMNHKNSLTFPDDNDISKEAKNLICAFLTDREVRLGRNGVEEIKRHLFFKNDQWAWE TLRDTVAPVVPDLSSDIDTSNFDDLEEDKGEEETFPIPKAFVGNQLPFVGFTYYSNRRYLSSANPNDNRTSSNADKSLQESLQKTIYKLE EQLHNEMQLKDEMEQKCRTSNIKLDKIMKELDEEGNQRRNLESTVSQIEKEKMLLQHRINEYQRKAEQENEKRRNVENEVSTLKDQLEDL KKVSQNSQLANEKLSQLQKQLEEANDLLRTESDTAVRLRKSHTEMSKSISQLESLNRELQERNRILENSKSQTDKDYYQLQAILEAERRD RGHDSEMIGDLQARITSLQEEVKHLKHNLEKVEGERKEAQDMLNHSEKEKNNLEIDLNYKLKSLQQRLEQEVNEHKVTKARLTDKHQSIE EAKSVAMCEMEKKLKEEREAREKAENRVVQIEKQCSMLDVDLKQSQQKLEHLTGNKERMEDEVKNLTLQLEQESNKRLLLQNELKTQAFE ADNLKGLEKQMKQEINTLLEAKRLLEFELAQLTKQYRGNEGQMRELQDQLEAEQYFSTLYKTQVKELKEEIEEKNRENLKKIQELQNEKE TLATQLDLAETKAESEQLARGLLEEQYFELTQESKKAASRNRQEITDKDHTVSRLEEANSMLTKDIEILRRENEELTEKMKKAEEEYKLE KEEEISNLKAAFEKNINTERTLKTQAVNKLAEIMNRKDFKIDRKKANTQDLRKKEKENRKLQLELNQEREKFNQMVVKHQKELNDMQAQL VEECAHRNELQMQLASKESDIEQLRAKLLDLSDSTSVASFPSADETDGNLPGSSSACDPAGTINSNLGYCQCKLHVEGPTCSRCKLLYWN LDKENPSGCSECKCHKAGTVSGTGECRQGDGDCHCKSHVGGDSCDTCEDGYFALEKSNYFGCQGCQCDIGGALSSMCSGPSGVCQCREHV VGKVCQRPENNYYFPDLHHMKYEIEDGSTPNGRDLRFGFDPLAFPEFSWRGYAQMTSVQNDVRITLNVGKSSGSLFRVILRYVNPGTEAV SGHITIYPSWGAAQSKEIIFLPSKEPAFVTVPGNGFADPFSITPGIWVACIKAEGVLLDYLVLLPRDYYEASVLQLPVTEPCAYAGPPQE NCLLYQHLPVTRFPCTLACEARHFLLDGEPRPVAVRQPTPAHPVMVDLSGREVELHLRLRIPQVGHYVVVVEYSTEAAQLFVVDVNVKSS GSVLAGQVNIYSCNYSVLCRSAVIDHMSRIAMYELLADADIQLKGHMARFLLHQVCIIPIEEFSAEYVRPQVHCIASYGRFVNQSATCVS LAHETPPTALILDVLSGRPFPHLPQQSSPSVDVLPGVTLKAPQNQVTLRGRVPHLGRYVFVIHFYQAAHPTFPAQVSVDGGWPRAGSFHA SFCPHVLGCRDQVIAEGQIEFDISEPEVAATVKVPEGKSLVLVRVLVVPAENYDYQILHKKSMDKSLEFITNCGKNSFYLDPQTASRFCK NSARSLVAFYHKGALPCECHPTGATGPHCSPEGGQCPCQPNVIGRQCTRCATGHYGFPRCKPCSCGRRLCEEMTGQCRCPPRTVRPQCEV CETHSFSFHPMAGCEGCNCSRRGTIEAAMPECDRDSGQCRCKPRITGRQCDRCASGFYRFPECVPCNCNRDGTEPGVCDPGTGACLCKEN VEGTECNVCREGSFHLDPANLKGCTSCFCFGVNNQCHSSHKRRTKFVDMLGWHLETADRVDIPVSFNPGSNSMVADLQELPATIHSASWV APTSYLGDKVSSYGGYLTYQAKSFGLPGDMVLLEKKPDVQLTGQHMSIIYEETNTPRPDRLHHGRVHVVEGNFRHASSRAPVSREELMTV LSRLADVRIQGLYFTETQRLTLSEVGLEEASDTGSGRIALAVEICACPPAYAGDSCQGCSPGYYRDHKGLYTGRCVPCNCNGHSNQCQDG SGICVNCQHNTAGEHCERCQEGYYGNAVHGSCRACPCPHTNSFATGCVVNGGDVRCSCKAGYTGTQCERCAPGYFGNPQKFGGSCQPCSC NSNGQLGSCHPLTGDCINQEPKDSSPAEECDDCDSCVMTLLNDLATMGEQLRLVKSQLQGLSASAGLLEQMRHMETQAKDLRNQLLNYRS AISNHGSKIEGLERELTDLNQEFETLQEKAQVNSRKAQTLNNNVNRATQSAKELDVKIKNVIRNVHILLKQISGTDGEGNNVPSGDFSRE WAEAQRMMRELRNRNFGKHLREAEADKRESQLLLNRIRTWQKTHQGENNGLANSIRDSLNEYEAKLSDLRARLQEAAAQAKQANGLNQEN ERALGAIQRQVKEINSLQSDFTKYLTTADSSLLQTNIALQLMEKSQKEYEKLAASLNEARQELSDKVRELSRSAGKTSLVEEAEKHARSL QELAKQLEEIKRNASGDELVRCAVDAATAYENILNAIKAAEDAANRAASASESALQTVIKEDLPRKAKTLSSNSDKLLNEAKMTQKKLKQ EVSPALNNLQQTLNIVTVQKEVIDTNLTTLRDGLHGIQRGDIDAMISSAKSMVRKANDITDEVLDGLNPIQTDVERIKDTYGRTQNEDFK KALTDADNSVNKLTNKLPDLWRKIESINQQLLPLGNISDNMDRIRELIQQARDAASKVAVPMRFNGKSGVEVRLPNDLEDLKGYTSLSLF LQRPNSRENGGTENMFVMYLGNKDASRDYIGMAVVDGQLTCVYNLGDREAELQVDQILTKSETKEAVMDRVKFQRIYQFARLNYTKGATS SKPETPGVYDMDGRNSNTLLNLDPENVVFYVGGYPPDFKLPSRLSFPPYKGCIELDDLNENVLSLYNFKKTFNLNTTEVEPCRRRKEESD KNYFEGTGYARVPTQPHAPIPTFGQTIQTTVDRGLLFFAENGDRFISLNIEDGKLMVRYKLNSELPKERGVGDAINNGRDHSIQIKIGKL QKRMWINVDVQNTIIDGEVFDFSTYYLGGIPIAIRERFNISTPAFRGCMKNLKKTSGVVRLNDTVGVTKKCSEDWKLVRSASFSRGGQLS FTDLGLPPTDHLQASFGFQTFQPSGILLDHQTWTRNLQVTLEDGYIELSTSDSGSPIFKSPQTYMDGLLHYVSVISDNSGLRLLIDDQLL RNSKRLKHISSSRQSLRLGGSNFEGCISNVFVQRLSLSPEVLDLTSNSLKRDVSLGGCSLNKPPFLMLLKGSTRFNKTKTFRINQLLQDT PVASPRSVKVWQDACSPLPKTQANHGALQFGDIPTSHLLFKLPQELLKPRSQFAVDMQTTSSRGLVFHTGTKNSFMALYLSKGRLVFALG TDGKKLRIKSKEKCNDGKWHTVVFGHDGEKGRLVVDGLRAREGSLPGNSTISIRAPVYLGSPPSGKPKSLPTNSFVGCLKNFQLDSKPLY TPSSSFGVSSCLGGPLEKGIYFSEEGGHVVLAHSVLLGPEFKLVFSIRPRSLTGILIHIGSQPGKHLCVYLEAGKVTASMDSGAGGTSTS VTPKQSLCDGQWHSVAVTIKQHILHLELDTDSSYTAGQIPFPPASTQEPLHLGGAPANLTTLRIPVWKSFFGCLRNIHVNHIPVPVTEAL -------------------------------------------------------------- |
Multiple Sequence Alignment of All Fusion Protein Isoforms |
Top |
Kinase Fusion Protein Functional Features |
 Four levels of functional features of fusion genes Four levels of functional features of fusion genesGo to FGviewer search page for the most frequent breakpoint (https://ccsmweb.uth.edu/FGviewer/chr18:18546878/chr18:21393021) - FGviewer provides the online visualization of the retention search of the protein functional features across DNA, RNA, protein, and pathological levels. - How to search 1. Put your fusion gene symbol. 2. Press the tab key until there will be shown the breakpoint information filled. 4. Go down and press 'Search' tab twice. 4. Go down to have the hyperlink of the search result. 5. Click the hyperlink. 6. See the FGviewer result for your fusion gene. |
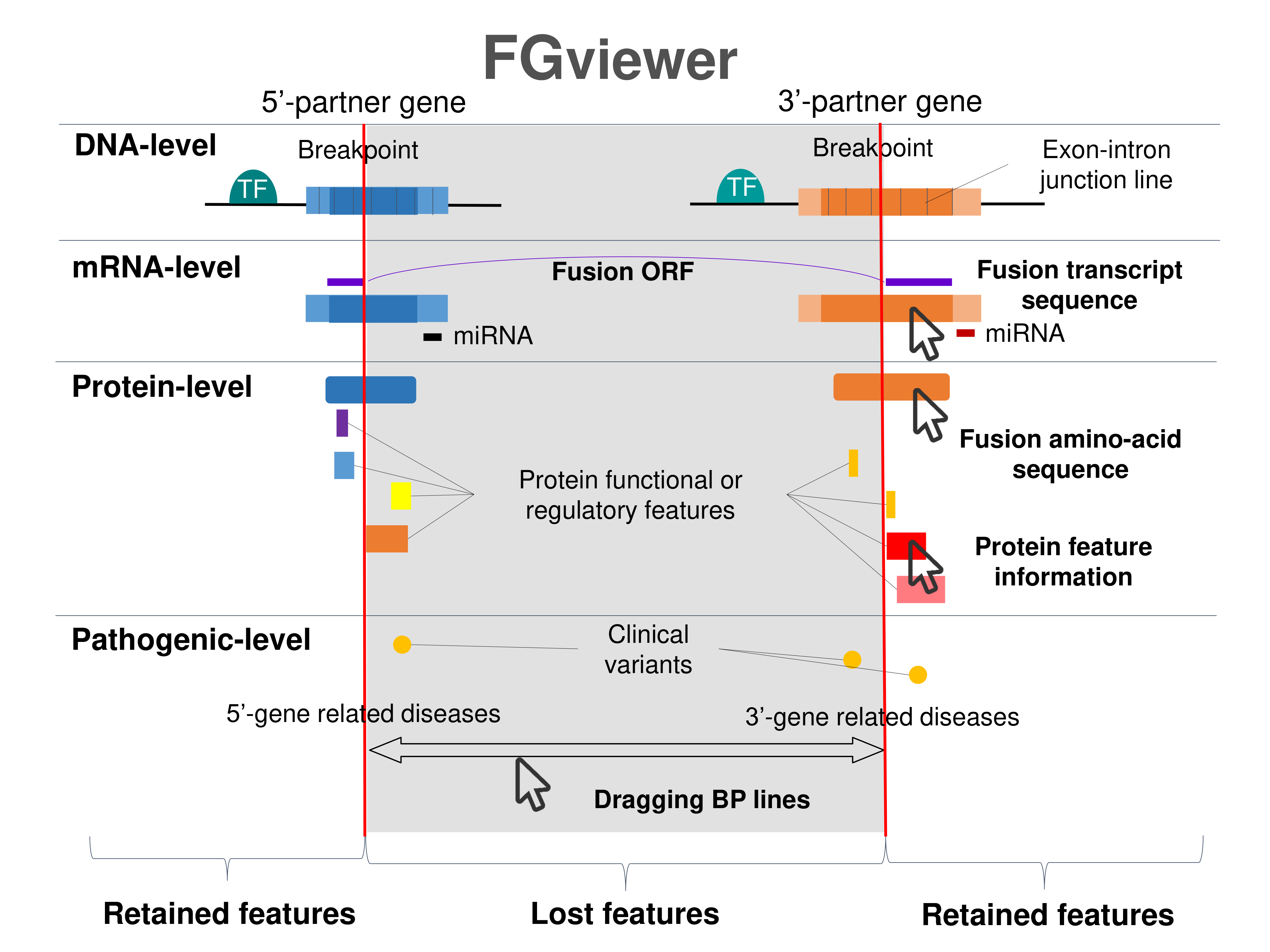 |
 Main function of each fusion partner protein. (from UniProt) Main function of each fusion partner protein. (from UniProt) |
| Hgene | Tgene |
| ROCK1 | LAMA3 |
| FUNCTION: Protein kinase which is a key regulator of the actin cytoskeleton and cell polarity (PubMed:10436159, PubMed:10652353, PubMed:11018042, PubMed:11283607, PubMed:17158456, PubMed:18573880, PubMed:19131646, PubMed:8617235, PubMed:9722579). Involved in regulation of smooth muscle contraction, actin cytoskeleton organization, stress fiber and focal adhesion formation, neurite retraction, cell adhesion and motility via phosphorylation of DAPK3, GFAP, LIMK1, LIMK2, MYL9/MLC2, TPPP, PFN1 and PPP1R12A (PubMed:10436159, PubMed:10652353, PubMed:11018042, PubMed:11283607, PubMed:17158456, PubMed:18573880, PubMed:19131646, PubMed:8617235, PubMed:9722579, PubMed:23093407, PubMed:23355470). Phosphorylates FHOD1 and acts synergistically with it to promote SRC-dependent non-apoptotic plasma membrane blebbing (PubMed:18694941). Phosphorylates JIP3 and regulates the recruitment of JNK to JIP3 upon UVB-induced stress (PubMed:19036714). Acts as a suppressor of inflammatory cell migration by regulating PTEN phosphorylation and stability (By similarity). Acts as a negative regulator of VEGF-induced angiogenic endothelial cell activation (PubMed:19181962). Required for centrosome positioning and centrosome-dependent exit from mitosis (By similarity). Plays a role in terminal erythroid differentiation (PubMed:21072057). Inhibits podocyte motility via regulation of actin cytoskeletal dynamics and phosphorylation of CFL1 (By similarity). Promotes keratinocyte terminal differentiation (PubMed:19997641). Involved in osteoblast compaction through the fibronectin fibrillogenesis cell-mediated matrix assembly process, essential for osteoblast mineralization (By similarity). May regulate closure of the eyelids and ventral body wall by inducing the assembly of actomyosin bundles (By similarity). {ECO:0000250|UniProtKB:P70335, ECO:0000250|UniProtKB:Q8MIT6, ECO:0000269|PubMed:10436159, ECO:0000269|PubMed:10652353, ECO:0000269|PubMed:11018042, ECO:0000269|PubMed:11283607, ECO:0000269|PubMed:17158456, ECO:0000269|PubMed:18573880, ECO:0000269|PubMed:18694941, ECO:0000269|PubMed:19036714, ECO:0000269|PubMed:19131646, ECO:0000269|PubMed:19181962, ECO:0000269|PubMed:19997641, ECO:0000269|PubMed:21072057, ECO:0000269|PubMed:23093407, ECO:0000269|PubMed:23355470, ECO:0000269|PubMed:8617235, ECO:0000269|PubMed:9722579}. | FUNCTION: Binding to cells via a high affinity receptor, laminin is thought to mediate the attachment, migration and organization of cells into tissues during embryonic development by interacting with other extracellular matrix components.; FUNCTION: Laminin-5 is thought to be involved in (1) cell adhesion via integrin alpha-3/beta-1 in focal adhesion and integrin alpha-6/beta-4 in hemidesmosomes, (2) signal transduction via tyrosine phosphorylation of pp125-FAK and p80, (3) differentiation of keratinocytes. |
 Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. Retention analysis result of each fusion partner protein across 39 protein features of UniProt such as six molecule processing features, 13 region features, four site features, six amino acid modification features, two natural variation features, five experimental info features, and 3 secondary structure features. Here, because of limited space for viewing, we only show the protein feature retention information belong to the 13 regional features. All retention annotation result can be downloaded at * Minus value of BPloci means that the break pointn is located before the CDS. |
 - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 5'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
| Hgene | ROCK1 | 18546878 | LAMA3 | 21393021 | ENST00000399799 | 27 | 33 | 341_409 | 11171 | 1355 | Domain | Note=AGC-kinase C-terminal;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00618 |
| Hgene | ROCK1 | 18546878 | LAMA3 | 21393021 | ENST00000399799 | 27 | 33 | 76_338 | 11171 | 1355 | Domain | Note=Protein kinase;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU00159 |
| Hgene | ROCK1 | 18546878 | LAMA3 | 21393021 | ENST00000399799 | 27 | 33 | 479_556 | 11171 | 1355 | Domain | Note=REM-1;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU01207 |
| Hgene | ROCK1 | 18546878 | LAMA3 | 21393021 | ENST00000399799 | 27 | 33 | 949_1015 | 11171 | 1355 | Domain | Note=RhoBD;Ontology_term=ECO:0000255;evidence=ECO:0000255|PROSITE-ProRule:PRU01206 |
 - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). - Retained domain in the 3'-partner of fusion protein (protein functional feature from UniProt). |
| Partner | Hgeneene | Hbp | Tgeneene | Tbp | ENST | BPexon | TotalExon | Protein feature loci | BPloci | TotalLen | Feature | Note |
Top |
Kinase Fusion Protein Structures |
 CIF files of the predicted kinase fusion proteins CIF files of the predicted kinase fusion proteins * Here we show the 3D structure of the fusion proteins using Mol*. AlphaFold produces a per-residue confidence score (pLDDT) between 0 and 100. Model confidence is shown from the pLDDT values per residue. pLDDT corresponds to the model’s prediction of its score on the local Distance Difference Test. It is a measure of local accuracy (from AlphfaFold website). To color code individual residues, we transformed individual PDB files into CIF format. |
| Kinase Fusion protein CIF link (fusion AA seq ID in KinaseFusionDB) | Henst | Tenst | Hgene | Hchr | Hbp | Tgene | Tchr | Tbp | AA seq | Len(AA seq) |
| PDB file >>>234_ROCK1_LAMA3 | ENST00000399799 | ENST00000313654 | ROCK1 | chr18 | 18546878 | LAMA3 | chr18 | 21393021 | MLVLVRGHCHSSSNMSTGDSFETRFEKMDNLLRDPKSEVNSDCLLDGLDALVYDLDFPALRKNKNIDNFLSRYKDTINKIRDLRMKAEDY EVVKVIGRGAFGEVQLVRHKSTRKVYAMKLLSKFEMIKRSDSAFFWEERDIMAFANSPWVVQLFYAFQDDRYLYMVMEYMPGGDLVNLMS NYDVPEKWARFYTAEVVLALDAIHSMGFIHRDVKPDNMLLDKSGHLKLADFGTCMKMNKEGMVRCDTAVGTPDYISPEVLKSQGGDGYYG RECDWWSVGVFLYEMLVGDTPFYADSLVGTYSKIMNHKNSLTFPDDNDISKEAKNLICAFLTDREVRLGRNGVEEIKRHLFFKNDQWAWE TLRDTVAPVVPDLSSDIDTSNFDDLEEDKGEEETFPIPKAFVGNQLPFVGFTYYSNRRYLSSANPNDNRTSSNADKSLQESLQKTIYKLE EQLHNEMQLKDEMEQKCRTSNIKLDKIMKELDEEGNQRRNLESTVSQIEKEKMLLQHRINEYQRKAEQENEKRRNVENEVSTLKDQLEDL KKVSQNSQLANEKLSQLQKQLEEANDLLRTESDTAVRLRKSHTEMSKSISQLESLNRELQERNRILENSKSQTDKDYYQLQAILEAERRD RGHDSEMIGDLQARITSLQEEVKHLKHNLEKVEGERKEAQDMLNHSEKEKNNLEIDLNYKLKSLQQRLEQEVNEHKVTKARLTDKHQSIE EAKSVAMCEMEKKLKEEREAREKAENRVVQIEKQCSMLDVDLKQSQQKLEHLTGNKERMEDEVKNLTLQLEQESNKRLLLQNELKTQAFE ADNLKGLEKQMKQEINTLLEAKRLLEFELAQLTKQYRGNEGQMRELQDQLEAEQYFSTLYKTQVKELKEEIEEKNRENLKKIQELQNEKE TLATQLDLAETKAESEQLARGLLEEQYFELTQESKKAASRNRQEITDKDHTVSRLEEANSMLTKDIEILRRENEELTEKMKKAEEEYKLE KEEEISNLKAAFEKNINTERTLKTQAVNKLAEIMNRKDFKIDRKKANTQDLRKKEKENRKLQLELNQEREKFNQMVVKHQKELNDMQAQL VEECAHRNELQMQLASKESDIEQLRAKLLDLSDSTSVASFPSADETDGNLPGSSSACDPAGTINSNLGYCQCKLHVEGPTCSRCKLLYWN LDKENPSGCSECKCHKAGTVSGTGECRQGDGDCHCKSHVGGDSCDTCEDGYFALEKSNYFGCQGCQCDIGGALSSMCSGPSGVCQCREHV VGKVCQRPENNYYFPDLHHMKYEIEDGSTPNGRDLRFGFDPLAFPEFSWRGYAQMTSVQNDVRITLNVGKSSGSLFRVILRYVNPGTEAV SGHITIYPSWGAAQSKEIIFLPSKEPAFVTVPGNGFADPFSITPGIWVACIKAEGVLLDYLVLLPRDYYEASVLQLPVTEPCAYAGPPQE NCLLYQHLPVTRFPCTLACEARHFLLDGEPRPVAVRQPTPAHPVMVDLSGREVELHLRLRIPQVGHYVVVVEYSTEAAQLFVVDVNVKSS GSVLAGQVNIYSCNYSVLCRSAVIDHMSRIAMYELLADADIQLKGHMARFLLHQVCIIPIEEFSAEYVRPQVHCIASYGRFVNQSATCVS LAHETPPTALILDVLSGRPFPHLPQQSSPSVDVLPGVTLKAPQNQVTLRGRVPHLGRYVFVIHFYQAAHPTFPAQVSVDGGWPRAGSFHA SFCPHVLGCRDQVIAEGQIEFDISEPEVAATVKVPEGKSLVLVRVLVVPAENYDYQILHKKSMDKSLEFITNCGKNSFYLDPQTASRFCK NSARSLVAFYHKGALPCECHPTGATGPHCSPEGGQCPCQPNVIGRQCTRCATGHYGFPRCKPCSCGRRLCEEMTGQCRCPPRTVRPQCEV CETHSFSFHPMAGCEGCNCSRRGTIEAAMPECDRDSGQCRCKPRITGRQCDRCASGFYRFPECVPCNCNRDGTEPGVCDPGTGACLCKEN VEGTECNVCREGSFHLDPANLKGCTSCFCFGVNNQCHSSHKRRTKFVDMLGWHLETADRVDIPVSFNPGSNSMVADLQELPATIHSASWV APTSYLGDKVSSYGGYLTYQAKSFGLPGDMVLLEKKPDVQLTGQHMSIIYEETNTPRPDRLHHGRVHVVEGNFRHASSRAPVSREELMTV LSRLADVRIQGLYFTETQRLTLSEVGLEEASDTGSGRIALAVEICACPPAYAGDSCQGCSPGYYRDHKGLYTGRCVPCNCNGHSNQCQDG SGICVNCQHNTAGEHCERCQEGYYGNAVHGSCRACPCPHTNSFATGCVVNGGDVRCSCKAGYTGTQCERCAPGYFGNPQKFGGSCQPCSC NSNGQLGSCHPLTGDCINQEPKDSSPAEECDDCDSCVMTLLNDLATMGEQLRLVKSQLQGLSASAGLLEQMRHMETQAKDLRNQLLNYRS AISNHGSKIEGLERELTDLNQEFETLQEKAQVNSRKAQTLNNNVNRATQSAKELDVKIKNVIRNVHILLKQISGTDGEGNNVPSGDFSRE WAEAQRMMRELRNRNFGKHLREAEADKRESQLLLNRIRTWQKTHQGENNGLANSIRDSLNEYEAKLSDLRARLQEAAAQAKQANGLNQEN ERALGAIQRQVKEINSLQSDFTKYLTTADSSLLQTNIALQLMEKSQKEYEKLAASLNEARQELSDKVRELSRSAGKTSLVEEAEKHARSL QELAKQLEEIKRNASGDELVRCAVDAATAYENILNAIKAAEDAANRAASASESALQTVIKEDLPRKAKTLSSNSDKLLNEAKMTQKKLKQ EVSPALNNLQQTLNIVTVQKEVIDTNLTTLRDGLHGIQRGDIDAMISSAKSMVRKANDITDEVLDGLNPIQTDVERIKDTYGRTQNEDFK KALTDADNSVNKLTNKLPDLWRKIESINQQLLPLGNISDNMDRIRELIQQARDAASKVAVPMRFNGKSGVEVRLPNDLEDLKGYTSLSLF LQRPNSRENGGTENMFVMYLGNKDASRDYIGMAVVDGQLTCVYNLGDREAELQVDQILTKSETKEAVMDRVKFQRIYQFARLNYTKGATS SKPETPGVYDMDGRNSNTLLNLDPENVVFYVGGYPPDFKLPSRLSFPPYKGCIELDDLNENVLSLYNFKKTFNLNTTEVEPCRRRKEESD KNYFEGTGYARVPTQPHAPIPTFGQTIQTTVDRGLLFFAENGDRFISLNIEDGKLMVRYKLNSELPKERGVGDAINNGRDHSIQIKIGKL QKRMWINVDVQNTIIDGEVFDFSTYYLGGIPIAIRERFNISTPAFRGCMKNLKKTSGVVRLNDTVGVTKKCSEDWKLVRSASFSRGGQLS FTDLGLPPTDHLQASFGFQTFQPSGILLDHQTWTRNLQVTLEDGYIELSTSDSGSPIFKSPQTYMDGLLHYVSVISDNSGLRLLIDDQLL RNSKRLKHISSSRQSLRLGGSNFEGCISNVFVQRLSLSPEVLDLTSNSLKRDVSLGGCSLNKPPFLMLLKGSTRFNKTKTFRINQLLQDT PVASPRSVKVWQDACSPLPKTQANHGALQFGDIPTSHLLFKLPQELLKPRSQFAVDMQTTSSRGLVFHTGTKNSFMALYLSKGRLVFALG TDGKKLRIKSKEKCNDGKWHTVVFGHDGEKGRLVVDGLRAREGSLPGNSTISIRAPVYLGSPPSGKPKSLPTNSFVGCLKNFQLDSKPLY TPSSSFGVSSCLGGPLEKGIYFSEEGGHVVLAHSVLLGPEFKLVFSIRPRSLTGILIHIGSQPGKHLCVYLEAGKVTASMDSGAGGTSTS VTPKQSLCDGQWHSVAVTIKQHILHLELDTDSSYTAGQIPFPPASTQEPLHLGGAPANLTTLRIPVWKSFFGCLRNIHVNHIPVPVTEAL | 3884 |
| 3D view using mol* of 234_ROCK1_LAMA3 | ||||||||||
| PDB file >>>HKFP_341_ROCK1_LAMA3 | ENST00000399799 | ENST00000313654 | ROCK1 | chr18 | 18546878 | LAMA3 | chr18 | 21393021 | MLVLVRGHCHSSSNMSTGDSFETRFEKMDNLLRDPKSEVNSDCLLDGLDALVYDLDFPALRKNKNIDNFLSRYKDTINKIRDLRMKAEDY EVVKVIGRGAFGEVQLVRHKSTRKVYAMKLLSKFEMIKRSDSAFFWEERDIMAFANSPWVVQLFYAFQDDRYLYMVMEYMPGGDLVNLMS NYDVPEKWARFYTAEVVLALDAIHSMGFIHRDVKPDNMLLDKSGHLKLADFGTCMKMNKEGMVRCDTAVGTPDYISPEVLKSQGGDGYYG RECDWWSVGVFLYEMLVGDTPFYADSLVGTYSKIMNHKNSLTFPDDNDISKEAKNLICAFLTDREVRLGRNGVEEIKRHLFFKNDQWAWE TLRDTVAPVVPDLSSDIDTSNFDDLEEDKGEEETFPIPKAFVGNQLPFVGFTYYSNRRYLSSANPNDNRTSSNADKSLQESLQKTIYKLE EQLHNEMQLKDEMEQKCRTSNIKLDKIMKELDEEGNQRRNLESTVSQIEKEKMLLQHRINEYQRKAEQENEKRRNVENEVSTLKDQLEDL KKVSQNSQLANEKLSQLQKQLEEANDLLRTESDTAVRLRKSHTEMSKSISQLESLNRELQERNRILENSKSQTDKDYYQLQAILEAERRD RGHDSEMIGDLQARITSLQEEVKHLKHNLEKVEGERKEAQDMLNHSEKEKNNLEIDLNYKLKSLQQRLEQEVNEHKVTKARLTDKHQSIE EAKSVAMCEMEKKLKEEREAREKAENRVVQIEKQCSMLDVDLKQSQQKLEHLTGNKERMEDEVKNLTLQLEQESNKRLLLQNELKTQAFE ADNLKGLEKQMKQEINTLLEAKRLLEFELAQLTKQYRGNEGQMRELQDQLEAEQYFSTLYKTQVKELKEEIEEKNRENLKKIQELQNEKE TLATQLDLAETKAESEQLARGLLEEQYFELTQESKKAASRNRQEITDKDHTVSRLEEANSMLTKDIEILRRENEELTEKMKKAEEEYKLE KEEEISNLKAAFEKNINTERTLKTQAVNKLAEIMNRKDFKIDRKKANTQDLRKKEKENRKLQLELNQEREKFNQMVVKHQKELNDMQAQL VEECAHRNELQMQLASKESDIEQLRAKLLDLSDSTSVASFPSADETDGNLPGSSSACDPAGTINSNLGYCQCKLHVEGPTCSRCKLLYWN LDKENPSGCSECKCHKAGTVSGTGECRQGDGDCHCKSHVGGDSCDTCEDGYFALEKSNYFGCQGCQCDIGGALSSMCSGPSGVCQCREHV VGKVCQRPENNYYFPDLHHMKYEIEDGSTPNGRDLRFGFDPLAFPEFSWRGYAQMTSVQNDVRITLNVGKSSGSLFRVILRYVNPGTEAV SGHITIYPSWGAAQSKEIIFLPSKEPAFVTVPGNGFADPFSITPGIWVACIKAEGVLLDYLVLLPRDYYEASVLQLPVTEPCAYAGPPQE NCLLYQHLPVTRFPCTLACEARHFLLDGEPRPVAVRQPTPAHPVMVDLSGREVELHLRLRIPQVGHYVVVVEYSTEAAQLFVVDVNVKSS GSVLAGQVNIYSCNYSVLCRSAVIDHMSRIAMYELLADADIQLKGHMARFLLHQVCIIPIEEFSAEYVRPQVHCIASYGRFVNQSATCVS LAHETPPTALILDVLSGRPFPHLPQQSSPSVDVLPGVTLKAPQNQVTLRGRVPHLGRYVFVIHFYQAAHPTFPAQVSVDGGWPRAGSFHA SFCPHVLGCRDQVIAEGQIEFDISEPEVAATVKVPEGKSLVLVRVLVVPAENYDYQILHKKSMDKSLEFITNCGKNSFYLDPQTASRFCK NSARSLVAFYHKGALPCECHPTGATGPHCSPEGGQCPCQPNVIGRQCTRCATGHYGFPRCKPCSCGRRLCEEMTGQCRCPPRTVRPQCEV CETHSFSFHPMAGCEGCNCSRRGTIEAAMPECDRDSGQCRCKPRITGRQCDRCASGFYRFPECVPCNCNRDGTEPGVCDPGTGACLCKEN VEGTECNVCREGSFHLDPANLKGCTSCFCFGVNNQCHSSHKRRTKFVDMLGWHLETADRVDIPVSFNPGSNSMVADLQELPATIHSASWV APTSYLGDKVSSYGGYLTYQAKSFGLPGDMVLLEKKPDVQLTGQHMSIIYEETNTPRPDRLHHGRVHVVEGNFRHASSRAPVSREELMTV LSRLADVRIQGLYFTETQRLTLSEVGLEEASDTGSGRIALAVEICACPPAYAGDSCQGCSPGYYRDHKGLYTGRCVPCNCNGHSNQCQDG SGICVNCQHNTAGEHCERCQEGYYGNAVHGSCRACPCPHTNSFATGCVVNGGDVRCSCKAGYTGTQCERCAPGYFGNPQKFGGSCQPCSC NSNGQLGSCHPLTGDCINQEPKDSSPAEECDDCDSCVMTLLNDLATMGEQLRLVKSQLQGLSASAGLLEQMRHMETQAKDLRNQLLNYRS AISNHGSKIEGLERELTDLNQEFETLQEKAQVNSRKAQTLNNNVNRATQSAKELDVKIKNVIRNVHILLKQISGTDGEGNNVPSGDFSRE WAEAQRMMRELRNRNFGKHLREAEADKRESQLLLNRIRTWQKTHQGENNGLANSIRDSLNEYEAKLSDLRARLQEAAAQAKQANGLNQEN ERALGAIQRQVKEINSLQSDFTKYLTTADSSLLQTNIALQLMEKSQKEYEKLAASLNEARQELSDKVRELSRSAGKTSLVEEAEKHARSL QELAKQLEEIKRNASGDELVRCAVDAATAYENILNAIKAAEDAANRAASASESALQTVIKEDLPRKAKTLSSNSDKLLNEAKMTQKKLKQ EVSPALNNLQQTLNIVTVQKEVIDTNLTTLRDGLHGIQRGDIDAMISSAKSMVRKANDITDEVLDGLNPIQTDVERIKDTYGRTQNEDFK KALTDADNSVNKLTNKLPDLWRKIESINQQLLPLGNISDNMDRIRELIQQARDAASKVAVPMRFNGKSGVEVRLPNDLEDLKGYTSLSLF LQRPNSRENGGTENMFVMYLGNKDASRDYIGMAVVDGQLTCVYNLGDREAELQVDQILTKSETKEAVMDRVKFQRIYQFARLNYTKGATS SKPETPGVYDMDGRNSNTLLNLDPENVVFYVGGYPPDFKLPSRLSFPPYKGCIELDDLNENVLSLYNFKKTFNLNTTEVEPCRRRKEESD KNYFEGTGYARVPTQPHAPIPTFGQTIQTTVDRGLLFFAENGDRFISLNIEDGKLMVRYKLNSELPKERGVGDAINNGRDHSIQIKIGKL QKRMWINVDVQNTIIDGEVFDFSTYYLGGIPIAIRERFNISTPAFRGCMKNLKKTSGVVRLNDTVGVTKKCSEDWKLVRSASFSRGGQLS FTDLGLPPTDHLQASFGFQTFQPSGILLDHQTWTRNLQVTLEDGYIELSTSDSGSPIFKSPQTYMDGLLHYVSVISDNSGLRLLIDDQLL RNSKRLKHISSSRQSLRLGGSNFEGCISNVFVQRLSLSPEVLDLTSNSLKRDVSLGGCSLNKPPFLMLLKGSTRFNKTKTFRINQLLQDT PVASPRSVKVWQDACSPLPKTQANHGALQFGDIPTSHLLFKLPQELLKPRSQFAVDMQTTSSRGLVFHTGTKNSFMALYLSKGRLVFALG TDGKKLRIKSKEKCNDGKWHTVVFGHDGEKGRLVVDGLRAREGSLPGNSTISIRAPVYLGSPPSGKPKSLPTNSFVGCLKNFQLDSKPLY TPSSSFGVSSCLGGPLEKGIYFSEEGGHVVLAHSVLLGPEFKLVFSIRPRSLTGILIHIGSQPGKHLCVYLEAGKVTASMDSGAGGTSTS VTPKQSLCDGQWHSVAVTIKQHILHLELDTDSSYTAGQIPFPPASTQEPLHLGGAPANLTTLRIPVWKSFFGCLRNIHVNHIPVPVTEAL | 3884_ROCK1_LAMA3 |
Top |
Comparison of Fusion Protein Isoforms |
 Superimpose the 3D Structures Among All Fusion Protein Isoforms Superimpose the 3D Structures Among All Fusion Protein Isoforms * Download the pdb file and open it from the molstar online viewer. |
 Comparison of the Secondary Structures of Fusion Protein Isoforms Comparison of the Secondary Structures of Fusion Protein Isoforms |
Top |
Comparison of Fusion Protein Sequences/Structures with Known Sequences/Structures from PDB |
Top |
pLDDT score distribution |
 pLDDT score distribution of the predicted fusion protein structures from AlphaFold2 pLDDT score distribution of the predicted fusion protein structures from AlphaFold2* AlphaFold produces a per-residue confidence score (pLDDT) between 0 and 100. * The blue color at the bottom marks the best active site residues. |
| 234_ROCK1_LAMA3.png |
 |
| 234_ROCK1_LAMA3.png |
 |
Top |
Potential Active Site Information |
 The potential binding sites of these fusion proteins were identified using SiteMap, a module of the Schrodinger suite. The potential binding sites of these fusion proteins were identified using SiteMap, a module of the Schrodinger suite. |
| Kinase Fusion AA seq ID in KinaseFusionDB | Site score | Size | Dscore | Volume | Exposure | Enclosure | Contact | Phobic | Philic | Balance | Don/Acc | Residues |
Top |
Ramachandran Plot of Kinase Fusion Protein Structure |
 Ramachandran plot of the torsional angles - phi (φ)and psi (ψ) - of the residues (amino acids) contained in this fusion protein peptide. Ramachandran plot of the torsional angles - phi (φ)and psi (ψ) - of the residues (amino acids) contained in this fusion protein peptide. |
| 234_ROCK1_LAMA3_ramachandran.png |
 |
Top |
Virtual Screening Results |
 Distribution of the average docking score across all approved kinase inhibitors. Distribution of the average docking score across all approved kinase inhibitors.Distribution of the number of occurrence across all approved kinase inhibitors. |
| 5'-kinase fusion protein case |
 |
| 3'-kinase fusion protein case |
Top |
 Drug information from DrugBank of the top 20 interacting small molecules. Drug information from DrugBank of the top 20 interacting small molecules.* The detailed information of individual kinase inhibitors are available in the download page. |
| Fusion gene name info | Drug | Docking score | Glide g score | Glide energy |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Neratinib | -8.4174 | -8.6033 | -71.4102 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Neratinib | -7.8147 | -7.997000000000001 | -68.1652 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Neratinib | -7.8147 | -7.997000000000001 | -68.1652 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Netarsudil | -7.789839999999999 | -7.80094 | -50.0937 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Netarsudil | -7.789839999999999 | -7.80094 | -50.0937 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Larotrectinib | -7.6759699999999995 | -7.6759699999999995 | -51.9208 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Crizotinib | -7.45335 | -7.94925 | -58.4495 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Crizotinib | -7.45335 | -7.94925 | -58.4495 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Larotrectinib | -7.24953 | -7.24953 | -51.7091 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Sunitinib | -7.22019 | -7.22439 | -42.4255 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Palbociclib | -7.203530000000001 | -7.61043 | -51.8243 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Palbociclib | -7.203530000000001 | -7.61043 | -51.8243 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Palbociclib | -7.1554 | -7.5623 | -51.7949 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Dacomitinib | -7.13972 | -7.2480199999999995 | -54.8939 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Dacomitinib | -7.13972 | -7.2480199999999995 | -54.8939 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Dacomitinib | -7.10032 | -7.20862 | -53.0718 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Dacomitinib | -7.10032 | -7.20862 | -53.0718 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Dacomitinib | -7.09962 | -7.20862 | -53.0718 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Cobimetinib | -7.09323 | -7.096030000000001 | -52.1278 |
| 234_ROCK1_LAMA3-DOCK_HTVS_1-001 | Axitinib | -7.0848 | -7.087999999999999 | -41.0847 |
Top |
Kinase-Substrate Information of ROCK1_LAMA3 |
 Phosphorylation target of the kinase Phosphorylation target of the kinase(phosphosite, 03-17-2024) |
| Kinase | Kinase UniProt Acc | Kinase species | Substrate | Substrate UniProt Acc | Substrate phosphorylated residues | Substrate phosphorylated sites (+/-7AA) | Domain |
| ROCK1 | Q13464 | human | NOS3 | P29474 | T495 | TGITRKKtFKEVANA | |
| ROCK1 | Q13464 | human | RDX | P35241 | T564 | AGRDKyKtLRQIRQG | ERM_C |
| ROCK1 | Q13464 | human | SLC9A1 | P19634 | T653 | LRsYNRHtLVADPyE | NEXCaM_BD |
| ROCK1 | Q13464 | human | ARHGAP35 | Q9NRY4 | T1173 | GRFASYRtsFsVGsD | RhoGAP_pG1_pG2 |
| ROCK1 | Q13464 | human | FHOD1 | Q9Y613 | S1137 | RsRGNRKsLRRtLKs | |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | S391 | RsSMNNGsPtALSGs | |
| ROCK1 | Q13464 | human | FHOD1 | Q9Y613 | T1141 | NRKsLRRtLKsGLGD | |
| ROCK1 | Q13464 | human | EZR | P15311 | T567 | QGRDkyKtLRQIRQG | ERM_C |
| ROCK1 | Q13464 | human | GFAP | P14136 | S38 | LGPGTRLsLARMPPP | Filament_head |
| ROCK1 | Q13464 | human | PTEN | P60484 | S229 | VkIYSsNsGPtRRED | PTEN_C2 |
| ROCK1 | Q13464 | human | ARHGAP35 | Q9NRY4 | S1236 | PKPKPRPsITKATWE | RhoGAP_pG1_pG2 |
| ROCK1 | Q13464 | human | DES | P17661 | T17 | RVssYRRtFGGAPGF | Filament_head |
| ROCK1 | Q13464 | human | AQP2 | P41181 | T269 | PQsLPRGtkA_____ | |
| ROCK1 | Q13464 | human | FHOD1 | Q9Y613 | S1131 | AARERKRsRGNRKsL | |
| ROCK1 | Q13464 | human | PRMT5 | O14744 | T80 | LsGRDWNtLIVGkLS | PRMT5_TIM |
| ROCK1 | Q13464 | human | RND3 | P61587 | S11 | RRAsQkLsSksIMDP | |
| ROCK1 | Q13464 | human | MYL9 | P24844 | T19 | KKRPQRAtsNVFAMF | |
| ROCK1 | Q13464 | human | DES | P17661 | S60 | VyQVsRtsGGAGGLG | Filament_head |
| ROCK1 | Q13464 | human | KCNK3 | O14649 | S336 | IPRDLStsDTCVEQS | |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | T452 | GLEKTQTtPNGSLQA | |
| ROCK1 | Q13464 | human | PFN1 | P07737 | S138 | MAsHLRRsQy_____ | Profilin |
| ROCK1 | Q13464 | human | RND3 | P61587 | S218 | QRATKRIsHMPsRPE | |
| ROCK1 | Q13464 | human | H3C1 | P68431 | S10 | tkQtArkstGGkAPr | Histone |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | S437 | SGIVTNGsFSSSNAE | |
| ROCK1 | Q13464 | human | RND3 | P61587 | S7 | _MKERRAsQkLsSks | |
| ROCK1 | Q13464 | human | LIMK2 | P53671 | T505 | NDRKKRYtVVGNPYW | PK_Tyr_Ser-Thr |
| ROCK1 | Q13464 | human | ARHGAP35 | Q9NRY4 | S1174 | RFASYRtsFsVGsDD | RhoGAP_pG1_pG2 |
| ROCK1 | Q13464 | human | H3C1 | P68431 | S28 | ATkAArksAPATGGV | Histone |
| ROCK1 | Q13464 | human | VIM | P08670 | S72 | ssAVrLrssVPGVRL | Filament_head |
| ROCK1 | Q13464 | human | GFAP | P14136 | S13 | ItsAArrsyVSsGEM | Filament_head |
| ROCK1 | Q13464 | human | DES | P17661 | T76 | LRAsRLGttRtPssy | Filament_head |
| ROCK1 | Q13464 | human | PTK2 | Q05397 | S732 | SsEGFYPsPQHMVQT | |
| ROCK1 | Q13464 | human | PAWR | Q96IZ0 | T163 | LREKRRstGVVNIPA | |
| ROCK1 | Q13464 | human | SOX9 | P48436 | S181 | YQPRRRKsVKNGQAE | |
| ROCK1 | Q13464 | human | PPP1R12A | O14974 | T853 | PREKRRstGVsFWtQ | |
| ROCK1 | Q13464 | human | PPP1R12A | O14974 | T696 | ARQsRRstQGVtLtD | |
| ROCK1 | Q13464 | human | MAPK8IP3 | Q9UPT6 | S365 | RLDRtGssPTQGIVN | |
| ROCK1 | Q13464 | human | SCRIB | Q14160 | S1508 | WRAARMksLEQDALR | |
| ROCK1 | Q13464 | human | MARCKS | P29966 | S159 | kkKKKRFsFkKsFkL | MARCKS |
| ROCK1 | Q13464 | human | RND3 | P61587 | S240 | LRKDKAKsCTVM___ | |
| ROCK1 | Q13464 | human | TPPP | O94811 | S107 | FSKIKGKsCRTITFE | p25-alpha |
| ROCK1 | Q13464 | human | GFAP | P14136 | T7 | _MERRRItsAArrsy | Filament_head |
| ROCK1 | Q13464 | human | ARHGAP35 | Q9NRY4 | S1150 | LERGRKVsIVsKPVL | RhoGAP_pG1_pG2 |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | S415 | HKLDVsRsPPLMVkk | |
| ROCK1 | Q13464 | human | ARHGAP19 | Q14CB8 | S422 | QkRARsRsFsGLIkR | |
| ROCK1 | Q13464 | human | MYL12B | O14950 | S20 | KRPQRAtsNVFAMFD | |
| ROCK1 | Q13464 | human | DPYSL2 | Q16555 | T555 | DNIPRRttQRIVAPP | |
| ROCK1 | Q13464 | human | TPPP | O94811 | S159 | sGVtKAIssPtVSRL | p25-alpha |
| ROCK1 | Q13464 | human | ARHGAP35 | Q9NRY4 | T1226 | LRsLRRNtKKPKPKP | RhoGAP_pG1_pG2 |
| ROCK1 | Q13464 | human | ADD1 | P35611 | T445 | QkQQREKtRWLNSGR | |
| ROCK1 | Q13464 | human | NR3C1 | P04150 | T519 | SENPGNKtIVPAtLP | |
| ROCK1 | Q13464 | human | MYL9 | P24844 | S20 | KRPQRAtsNVFAMFD | |
| ROCK1 | Q13464 | human | DES | P17661 | T77 | RAsRLGttRtPssyG | Filament_head |
| ROCK1 | Q13464 | human | ADD1 | P35611 | T480 | TKEDGHRtstsAVPN | |
| ROCK1 | Q13464 | human | SCRIB | Q14160 | S1378 | EGPPKRVsLVGADDL | |
| ROCK1 | Q13464 | human | MSN | P26038 | T558 | LGRDKyKtLRQIRQG | ERM_C |
| ROCK1 | Q13464 | human | NR3C1 | P04150 | S617 | WRSYRQSsANLLCFA | Hormone_recep |
| ROCK1 | Q13464 | human | PPP1R12B | O60237 | T646 | ARQtRRstQGVtLtD | |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | S413 | sVHKLDVsRsPPLMV | |
| ROCK1 | Q13464 | human | DAPK3 | O43293 | T265 | KDPKRRMtIAQSLEH | Pkinase |
| ROCK1 | Q13464 | human | MYL12B | O14950 | T19 | KKRPQRAtsNVFAMF | |
| ROCK1 | Q13464 | human | ARHGAP24 | Q8N264 | S402 | LSGsKTNsPKNsVHK | |
| ROCK1 | Q13464 | human | DES | P17661 | S12 | ysSSQRVssYRRtFG | Filament_head |
| ROCK1 | Q13464 | human | LIMK1 | P53667 | T508 | PDRKKRYtVVGNPYW | PK_Tyr_Ser-Thr |
| ROCK1 | Q13464 | human | DAPK3 | O43293 | T299 | PERRRLKtTRLkEyt | |
| ROCK1 | Q13464 | human | MAPK8IP3 | Q9UPT6 | S314 | RAREKRDsRNMEVQV | |
| ROCK1 | Q13464 | human | TPPP | O94811 | S32 | DRAAKRLsLEsEGAG | |
| ROCK1 | Q13464 | human | MAPK8IP3 | Q9UPT6 | S364 | TRLDRtGssPTQGIV | |
| ROCK1 | Q13464 | human | PTEN | P60484 | T232 | YSsNsGPtRREDKFM | PTEN_C2 |
| ROCK1 | Q13464 | human | KCNK3 | O14649 | S393 | GLMKRRssV______ | |
| ROCK1 | Q13464 | human | PPP1R12A | O14974 | S852 | RPREKRRstGVsFWt |
 Biological Network Integration of This Kinase and Substrates Biological Network Integration of This Kinase and Substrates (GeneMANIA website) |
 Enriched GO biological processes of the phosphorylation target genes of the kinase Enriched GO biological processes of the phosphorylation target genes of the kinase |
| Kinase | GOID | GO term | P.adjust |
| ROCK1 | ID | Description | 0.00e+00 |
| ROCK1 | GO:0051017 | actin filament bundle assembly | 1.38e-08 |
| ROCK1 | GO:0061572 | actin filament bundle organization | 1.38e-08 |
| ROCK1 | GO:0007015 | actin filament organization | 1.55e-08 |
| ROCK1 | GO:1902946 | protein localization to early endosome | 2.04e-06 |
| ROCK1 | GO:0008360 | regulation of cell shape | 2.21e-06 |
| ROCK1 | GO:0032956 | regulation of actin cytoskeleton organization | 3.56e-05 |
| ROCK1 | GO:0036010 | protein localization to endosome | 3.56e-05 |
| ROCK1 | GO:0042063 | gliogenesis | 3.56e-05 |
| ROCK1 | GO:0022604 | regulation of cell morphogenesis | 5.66e-05 |
| ROCK1 | GO:0032535 | regulation of cellular component size | 5.66e-05 |
| ROCK1 | GO:0010001 | glial cell differentiation | 5.66e-05 |
| ROCK1 | GO:0032970 | regulation of actin filament-based process | 5.66e-05 |
| ROCK1 | GO:1902903 | regulation of supramolecular fiber organization | 6.36e-05 |
| ROCK1 | GO:0110053 | regulation of actin filament organization | 7.54e-05 |
| ROCK1 | GO:0030953 | astral microtubule organization | 7.54e-05 |
| ROCK1 | GO:1905668 | positive regulation of protein localization to endosome | 1.29e-04 |
| ROCK1 | GO:0032231 | regulation of actin filament bundle assembly | 1.49e-04 |
| ROCK1 | GO:1905666 | regulation of protein localization to endosome | 1.49e-04 |
| ROCK1 | GO:0061028 | establishment of endothelial barrier | 1.75e-04 |
| ROCK1 | GO:1903651 | positive regulation of cytoplasmic transport | 2.07e-04 |
| ROCK1 | GO:0002064 | epithelial cell development | 2.07e-04 |
| ROCK1 | GO:2000641 | regulation of early endosome to late endosome transport | 3.81e-04 |
| ROCK1 | GO:0051893 | regulation of focal adhesion assembly | 3.81e-04 |
| ROCK1 | GO:0090109 | regulation of cell-substrate junction assembly | 3.81e-04 |
| ROCK1 | GO:0001885 | endothelial cell development | 3.90e-04 |
| ROCK1 | GO:0150116 | regulation of cell-substrate junction organization | 4.78e-04 |
| ROCK1 | GO:0043254 | regulation of protein-containing complex assembly | 6.69e-04 |
| ROCK1 | GO:0042060 | wound healing | 6.86e-04 |
| ROCK1 | GO:0048041 | focal adhesion assembly | 1.04e-03 |
| ROCK1 | GO:0048708 | astrocyte differentiation | 1.10e-03 |
| ROCK1 | GO:0008361 | regulation of cell size | 1.28e-03 |
| ROCK1 | GO:1900027 | regulation of ruffle assembly | 1.29e-03 |
| ROCK1 | GO:0051492 | regulation of stress fiber assembly | 1.30e-03 |
| ROCK1 | GO:0031345 | negative regulation of cell projection organization | 1.30e-03 |
| ROCK1 | GO:1903649 | regulation of cytoplasmic transport | 1.30e-03 |
| ROCK1 | GO:0007044 | cell-substrate junction assembly | 1.30e-03 |
| ROCK1 | GO:0120032 | regulation of plasma membrane bounded cell projection assembly | 1.59e-03 |
| ROCK1 | GO:0060491 | regulation of cell projection assembly | 1.59e-03 |
| ROCK1 | GO:0150115 | cell-substrate junction organization | 1.59e-03 |
| ROCK1 | GO:0031032 | actomyosin structure organization | 1.64e-03 |
| ROCK1 | GO:0110020 | regulation of actomyosin structure organization | 1.64e-03 |
| ROCK1 | GO:0044319 | wound healin | 4.74e-05 |
| ROCK1 | GO:0048709 | oligodendrocyte differentiation | 1.64e-03 |
| ROCK1 | GO:0090504 | epiboly | 1.70e-03 |
| ROCK1 | GO:1902115 | regulation of organelle assembly | 1.73e-03 |
| ROCK1 | GO:0030038 | contractile actin filament bundle assembly | 1.73e-03 |
| ROCK1 | GO:0043149 | stress fiber assembly | 1.73e-03 |
| ROCK1 | GO:0007163 | establishment or maintenance of cell polarity | 2.08e-03 |
| ROCK1 | GO:0045022 | early endosome to late endosome transport | 2.34e-03 |
Top |
Related Drugs to ROCK1_LAMA3 |
 Drugs used for this fusion-positive patient. Drugs used for this fusion-positive patient. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Drug | Source | PMID |
 Distribution of the number of studies mentioning ROCK1-LAMA3 and kinase inhibitors the PubMed Abstract (04-01-2024) Distribution of the number of studies mentioning ROCK1-LAMA3 and kinase inhibitors the PubMed Abstract (04-01-2024) |
| Fusion gene - drug pair 1 | Fusion gene - drug pair 2 | PMID | Publication date | DOI | Study title |
Top |
Related Diseases to ROCK1_LAMA3 |
 Diseases that have this fusion gene. Diseases that have this fusion gene. (Manual curation of PubMed, 04-30-2022 + MyCancerGenome) |
| Hgene | Tgene | Disease | Source | PMID |
 Related diseases from the literature mentioned this fusion gene and drug. Related diseases from the literature mentioned this fusion gene and drug. (PubMed, 04-01-2024) |
| MeSH ID | MeSH term |
 Diseases associated with fusion partners. Diseases associated with fusion partners. (DisGeNet 4.0) |
| Partner | Gene | Disease ID | Disease name | # pubmeds | Source |
| Tgene | LAMA3 | C1328355 | Laryngoonychocutaneous syndrome | 5 | CTD_human;GENOMICS_ENGLAND;ORPHANET |
| Tgene | LAMA3 | C0079683 | Herlitz Disease | 4 | CTD_human;GENOMICS_ENGLAND |
| Tgene | LAMA3 | C0079301 | Junctional Epidermolysis Bullosa | 3 | CTD_human;GENOMICS_ENGLAND |
| Tgene | LAMA3 | C0268374 | Adult junctional epidermolysis bullosa (disorder) | 3 | CTD_human;GENOMICS_ENGLAND |
Top |
Clinical Trials of the Found Drugs/Small Molecules |
 Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) Statistics of the Clinical Trials of the Found Kinase Inibitors from clinicaltrials.gov (06-17-2024) |
 Clinical Trials from clinicaltrials.gov (06-17-2024) Clinical Trials from clinicaltrials.gov (06-17-2024) |
| Fusion Gene | Kinase Inhibitor | NCT ID | Study Status | Phases | Disease | # Enrolment | Date |

